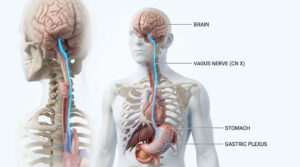
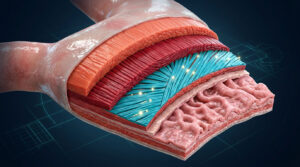
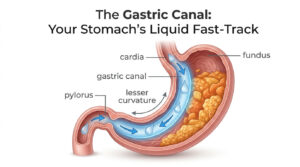
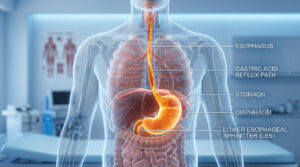
Oncology, a field of medical science, involves diagnosis and treatment of cancer. Cancer, which is caused due to uncontrolled cellular division, is one of the biggest healthcare threats in developed as well as developing nations, with genetics and lifestyle equally fueling the ‘killer’ condition. The causative factors for this condition are many and early and timely diagnosis is the only path for successful treatment and remission.