As a board-certified gastroenterologist with over 15 years of clinical experience, I hear one question more than almost any other. Patients walk into my office holding a prescription bottle, looking worried. The question is always the same: do antibiotics cause diarrhea?
Table of Contents
The short answer is yes, they absolutely can. Antibiotics save lives every day around the world, but they also cause real gastrointestinal problems that should not be brushed off. Antibiotic-associated diarrhea is more than a minor inconvenience. It reflects a complex disruption happening inside your gut, and in some cases, ignoring it can lead to serious, even life-threatening complications.
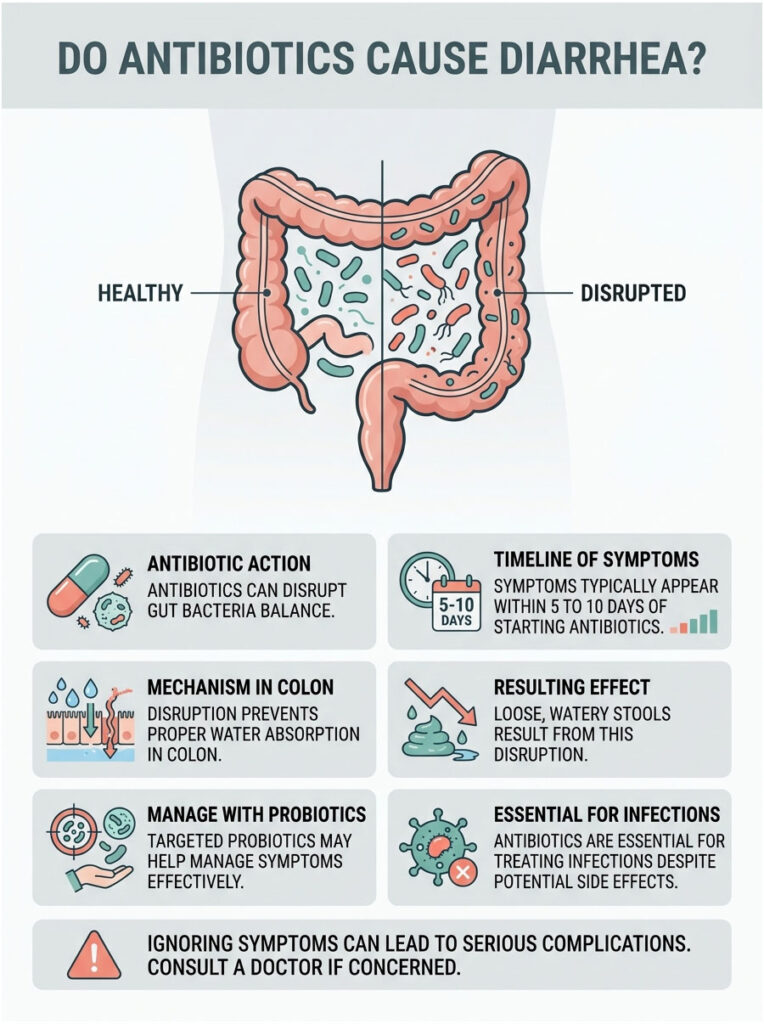
Quick Answer: Yes, antibiotics frequently cause diarrhea by wiping out the beneficial bacteria in your digestive tract. This disruption prevents your colon from absorbing water properly, leading to loose, watery stools. Symptoms typically show up within 5 to 10 days of starting your medication, but targeted probiotics can help manage them effectively.
Key Clinical Statistics
- Up to 35% of patients on antimicrobial therapy develop antibiotic-associated diarrhea.
- The CDC reports nearly 500,000 C. diff infections annually in the United States.
- Delayed-onset digestive symptoms can appear up to 8 weeks after finishing a course of antibiotics.
- Clinically studied probiotics can reduce the overall risk of diarrhea by more than 40%.
- Adults over 65 face a 10 times greater risk of severe complications from microbiome disruption.
- About 20% of patients who develop a C. diff infection will experience a recurrence within weeks.
Why Do Antibiotics Cause Diarrhea in Otherwise Healthy People?
The Hidden World Inside Your Gut
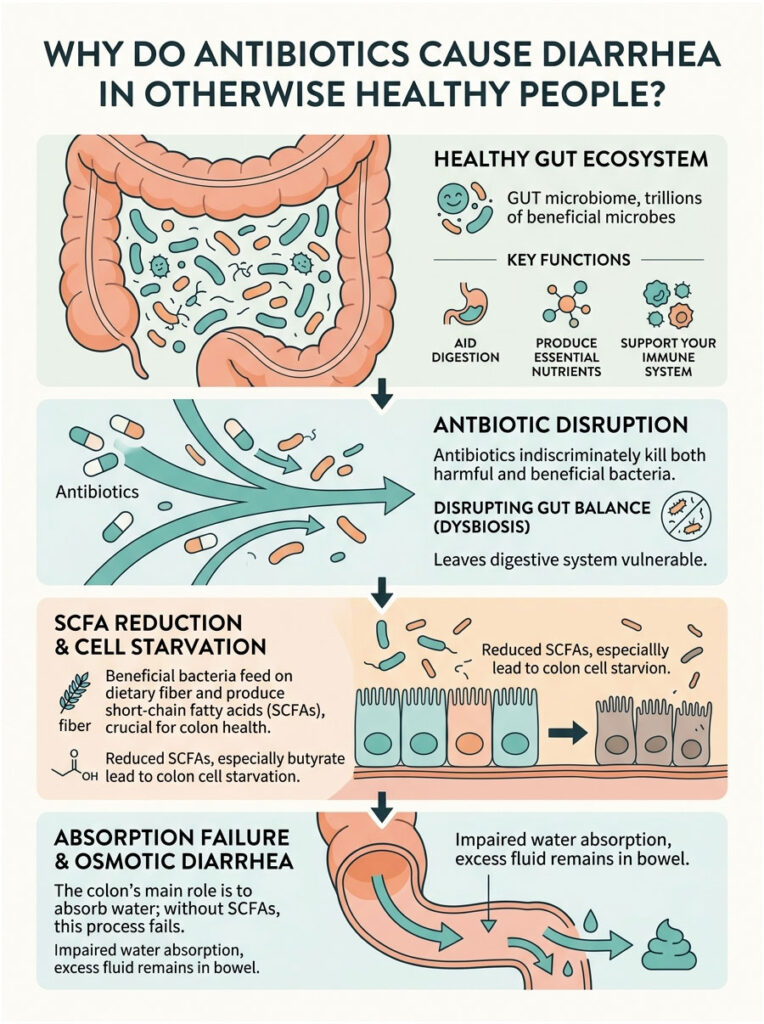
To understand why antibiotics upset your stomach, you first need to appreciate what lives inside you. Your digestive tract is home to tens of trillions of microscopic organisms. These microbes coexist in a finely tuned balance, helping you digest food every single day.
Doctors call these helpful organisms commensal bacteria. They do far more than just break down meals. They produce essential nutrients, synthesize vitamins, and train your immune system to fend off harmful invaders. Think of it like a dense, thriving rainforest inside your colon, where every species of bacteria plays a specific role. When this ecosystem stays balanced, your bowel movements remain regular and well-formed.
How Antibiotics Wage War on Your Gut Flora
Here is the uncomfortable truth about modern antibiotics. Most of them cannot tell the difference between the bacteria causing your infection and the good bacteria keeping your gut healthy. They work more like a bomb than a surgical strike.
When you swallow a pill, that medication travels through your entire digestive system before reaching the site of infection. Along the way, it causes massive collateral damage to your friendly commensal bacteria. This sudden imbalance in your gut microbiome is what clinicians call dysbiosis.
Dysbiosis changes the entire landscape of your intestines. The protective rainforest gets clear-cut, and your digestive tract is left exposed and unable to carry out its normal functions.
Short-Chain Fatty Acids and Colonic Starvation
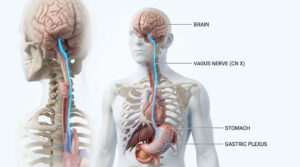
Let us look more closely at the biochemistry behind this. Your beneficial bacteria survive by feeding on dietary fiber from the foods you eat. In return, they produce chemical byproducts called short-chain fatty acids, or SCFAs. The most important of these is butyrate, which serves as the primary fuel source for the cells lining your colon walls.
When antibiotics kill off your good bacteria, butyrate production drops sharply. Without this fuel, your colon cells essentially begin to starve. A starving colon quickly loses its ability to do its main job.
The Mechanics of Osmotic Diarrhea
So what exactly is the colon’s main job? It absorbs water from digested food before waste leaves your body. When colon cells are starved of short-chain fatty acids, water absorption grinds to a halt.
All the liquid from the food and drinks you consume stays trapped in your bowel. This excess fluid mixes with stool and rushes through your system rapidly. In medical terms, this is called osmotic diarrhea.
Osmotic diarrhea is a direct, mechanical result of dysbiosis. It is not an allergic reaction to the drug itself. It is a physical failure of your intestines to absorb water because the bacteria responsible for powering that process have been wiped out.
Which Antibiotic Classes Cause Diarrhea Most Often?
Broad-Spectrum vs. Narrow-Spectrum Antibiotics
Not every antibiotic carries the same risk for your gut. In clinical practice, we group antibacterial drugs into two categories: broad-spectrum and narrow-spectrum.
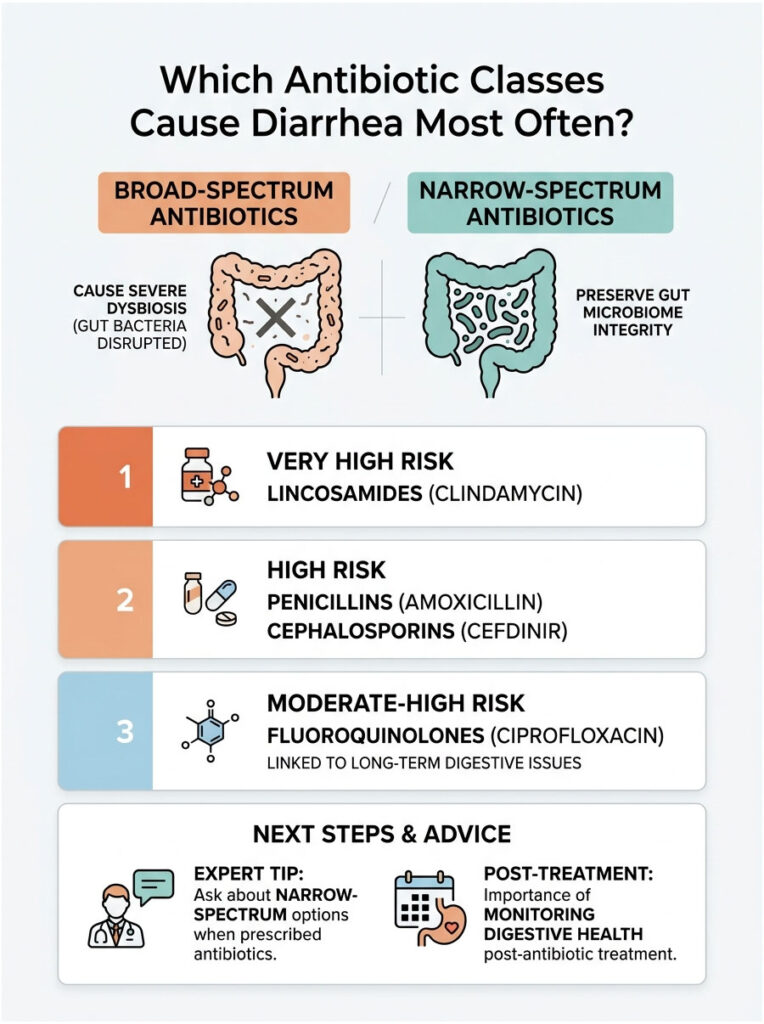
Broad-spectrum antibiotics are designed to kill a wide variety of bacterial strains at once. Doctors often prescribe them when they are not certain which specific bacterium is responsible for the illness. Because they attack so many different microbes, broad-spectrum drugs cause the most severe dysbiosis.
Narrow-spectrum agents, on the other hand, target only a few specific types of bacteria. They leave the vast majority of your gut microbiome intact, which drastically lowers your risk of antibiotic-associated diarrhea.
Expert Tip: When your doctor hands you a prescription, ask a simple question: “Is there a narrow-spectrum option for my specific infection?” This one conversation can save your gut microbiome from unnecessary damage.
High-Risk: Penicillins and Cephalosporins
Certain drug classes are well known in gastroenterology for causing severe digestive problems. Aminopenicillins are among the most common offenders. Amoxicillin and Amoxicillin-Clavulanate (sold as Augmentin) fall into this group.
Augmentin is heavily prescribed for stubborn ear infections, sinus infections, and bronchitis. Its powerful chemical combination alters gut flora rapidly, leading to increased bowel contractions and fast-onset osmotic diarrhea.
Cephalosporins are another high-risk category. Drugs like Cefdinir and Cephalexin are broad-spectrum agents that significantly reduce colonization resistance, wiping out large populations of healthy bacteria within days.
The Danger of Lincosamides
When it comes to severe microbiome disruption, Clindamycin stands out. It belongs to the Lincosamide class and is historically one of the worst offenders for gastrointestinal distress.
Dentists and dermatologists frequently prescribe Clindamycin for tooth infections and severe acne. Unfortunately, it heavily disrupts the gut ecosystem and carries an exceptionally high association with dangerous secondary bacterial infections. If your doctor prescribes it, pay close attention to your digestive health. The damage it causes to commensal bacteria can take months to repair.
Fluoroquinolones and Long-Term Damage
Fluoroquinolones carry some of the highest risks in modern pharmacology. You may recognize them by names like Ciprofloxacin and Levofloxacin. They are potent broad-spectrum antibiotics used for urinary tract infections and pneumonia.
The FDA has issued multiple black box warnings for fluoroquinolones. Beyond risks to tendons and the nervous system, these drugs cause deep, long-lasting dysbiosis. Patients often report digestive problems that persist for a year or longer. They eradicate such a wide range of bacteria that the microbiome struggles to naturally rebuild itself.
Antibiotic Classes and Diarrhea Risk: A Clinical Comparison
| Antibiotic Class | Common Examples | Risk Level | Notes on Microbiome Impact |
| Lincosamides | Clindamycin | Very High | Highest rates of C. difficile overgrowth and severe, long-term dysbiosis. |
| Aminopenicillins | Amoxicillin, Augmentin | High | Frequently prescribed. Alters gut flora rapidly, increasing bowel contractions. |
| Cephalosporins | Cefdinir, Cephalexin | High | Broad-spectrum action significantly reduces colonization resistance. |
| Fluoroquinolones | Ciprofloxacin, Levaquin | Moderate-High | Carries FDA black box warnings. Linked to delayed-onset and lasting dysbiosis. |
| Macrolides | Azithromycin (Z-Pak) | Moderate | Often causes hypermotility alongside mild to moderate bacterial destruction. |
| Tetracyclines | Doxycycline | Low-Moderate | Generally better tolerated with less impact on short-chain fatty acid production. |
Recognizing Clostridioides difficile (C. diff) Infection
How Your Commensal Bacteria Protect You
Standard osmotic diarrhea is frustrating, but there is a far more dangerous complication to watch for. It involves a highly aggressive bacterium called Clostridioides difficile, commonly known as C. diff.
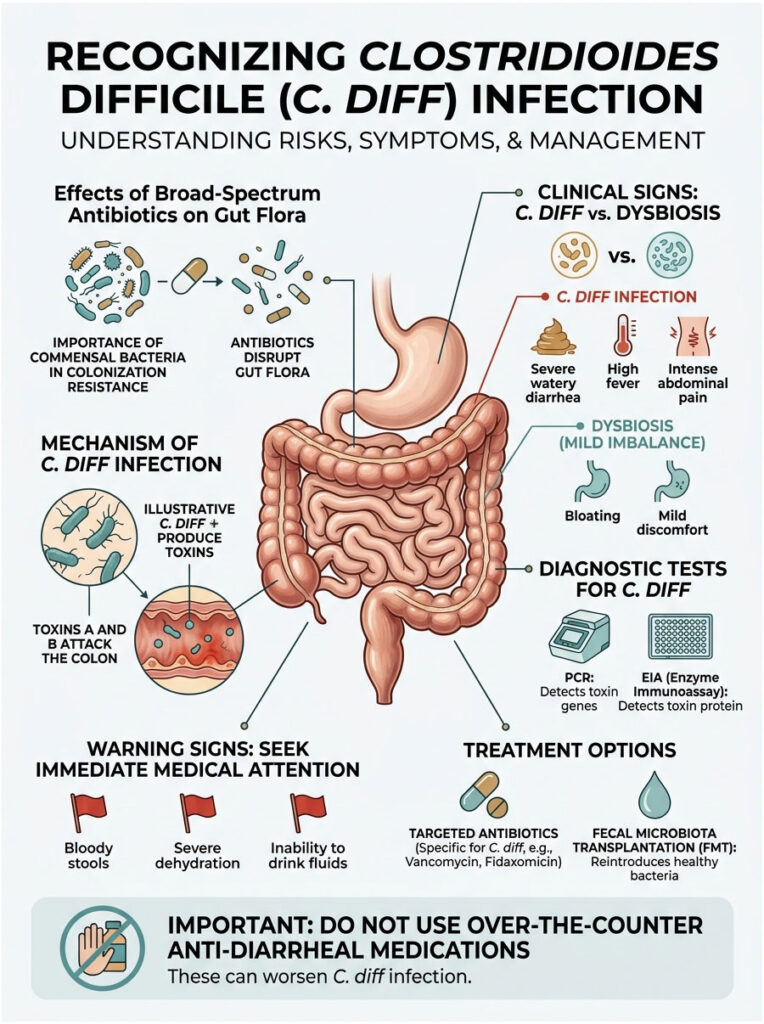
Healthy commensal bacteria provide a critical defense mechanism known as colonization resistance. They occupy all the physical space and consume all the available nutrients inside your colon, which prevents harmful invaders from gaining a foothold. When broad-spectrum antibiotics eliminate your good bacteria, this protective barrier vanishes entirely, and opportunistic pathogens seize the opportunity.
How C. diff Toxins Attack Your Colon
Without commensal bacteria keeping it in check, C. diff spores begin to germinate. They multiply rapidly in the emptied space of your colon and release two destructive poisons known as Toxin A and Toxin B.
These toxins directly attack the mucosal lining of your intestines, destroying the cellular junctions that hold your gut barrier together. The resulting damage causes severe, bleeding inflammation. Clinically, this condition is called pseudomembranous colitis. It is no longer just an upset stomach from a pill. It is a serious, active, and contagious bacterial infection of the colon.
Warning Signs That Demand Immediate Medical Attention
Knowing how to tell standard antibiotic-associated diarrhea apart from a C. diff infection could save your life. Standard dysbiosis usually involves mild to moderate loose stools without systemic illness or fever.
C. diff presents with urgent red flags. Seek medical attention immediately if you experience severe watery bowel movements more than five times a day, a high fever, intense abdominal cramping, or visible blood in your stool. Another distinctive sign is an unusually foul odor to the stool.
Important Warning: If you suspect C. diff, do not take over-the-counter anti-diarrheal medications like Imodium. These drugs slow bowel movement, which traps the dangerous toxins inside your colon and can worsen the infection.
How Doctors Diagnose C. diff
When we suspect a C. diff infection, we run specific lab tests without delay. Your doctor will request a liquid stool sample for analysis. The laboratory performs a Polymerase Chain Reaction (PCR) test to detect the bacterium’s genetic material and an Enzyme Immunoassay (EIA) to check whether the toxins are actively present. Both tests are needed for a definitive diagnosis.
One strict rule in clinical gastroenterology: you cannot test formed, solid stool for C. diff. The bacterium only causes active disease when stool is liquid and unformed.
Treating C. diff Infections
Treating C. diff involves a clinical irony. To fix an antibiotic-induced infection, doctors prescribe more antibiotics, but highly targeted ones that attack only C. diff. Oral Vancomycin and Fidaxomicin are the standard choices. These drugs stay entirely within your digestive tract instead of absorbing into the bloodstream, allowing your normal flora a chance to recover.
For patients suffering from multiple recurrences, doctors may use Fecal Microbiota Transplantation (FMT). This procedure transfers carefully screened, healthy donor stool into the patient’s colon, instantly restoring colonization resistance and resolving the dysbiosis.
How Long Does Antibiotic-Associated Diarrhea Last?

Early-Onset Symptoms
Patients often want to know exactly how long they will be dealing with these symptoms. The answer depends on when symptoms begin and how severely your microbiome was affected.
Early-onset symptoms typically start between 5 and 10 days after beginning your prescription. This makes biological sense, as it takes roughly a week for the medication to significantly reduce commensal bacteria populations. Once the bacterial population drops below a critical threshold, water absorption falters and bowel movements become loose and urgent.
Why You Should Never Stop Antibiotics Early
If early symptoms appear, do not stop taking your prescribed medication without consulting your doctor. Stopping a course of antibiotics prematurely leaves surviving bacteria to mutate and develop resistance, potentially creating dangerous superbugs that are far harder to treat later.
Instead, contact your healthcare provider right away. Doctors can often adjust the dosage, switch you to a narrow-spectrum alternative, or add targeted probiotics to manage side effects safely.
The Eight-Week Vulnerability Window
Many patients are surprised to develop digestive problems well after finishing their medication. Delayed-onset diarrhea is a well-documented clinical phenomenon. You remain vulnerable to dysbiosis for up to 8 weeks after swallowing your last pill.
The reason is straightforward. It takes significant time for gut flora to rebuild its defensive barriers. During this recovery window, short-chain fatty acids remain depleted and colonization resistance is practically nonexistent. This is exactly why someone can pick up a C. diff infection from a hospital surface a full month after finishing Amoxicillin.
Long-Term Microbiome Recovery
Even after standard osmotic diarrhea resolves in a few days, your internal ecosystem remains fragile. Recent research from the American Gastroenterological Association (AGA) shows that some patients never fully recover their original bacterial diversity. Certain rare strains may be lost permanently after a single course of broad-spectrum drugs.
This is why proactive recovery matters. You cannot simply wait for your gut to heal on its own. Actively rebuilding your microbiome through diet, hydration, and targeted supplementation is essential.
Can Probiotics Prevent Antibiotic-Associated Diarrhea?

Why Most Generic Supplements Fall Short
You do not have to accept digestive damage as inevitable. Using probiotics for prevention is a well-supported, evidence-based strategy. But there is an important nuance: not all probiotics are created equal.
Grabbing a random, inexpensive bottle off a grocery store shelf will likely accomplish nothing. Many commercial products contain weak strains that die the moment they encounter stomach acid. To get real protection, you need clinically studied, medical-grade strains with proven efficacy against antibiotic-associated diarrhea.
Saccharomyces boulardii: The Medicinal Yeast
In gastroenterology, one organism gets special attention: Saccharomyces boulardii. It is not a bacterium at all, but a highly beneficial medicinal yeast.
Because S. boulardii is a yeast, antibacterial medications cannot kill it. It is naturally immune to the drugs you are taking, which means it survives the medication course and travels safely into your colon. Once there, it secretes proteins that actively break down and neutralize C. diff toxins. It also stimulates your intestinal lining to produce more immune cells, shielding your vulnerable gut barrier.
Lactobacillus rhamnosus GG: The Barrier Builder
Another powerhouse strain is Lactobacillus rhamnosus GG, often labeled simply as LGG on supplement bottles. This bacterial strain excels at patching up microscopic gaps in the gut barrier.
LGG physically binds to intestinal walls, crowds out harmful bacteria, and encourages surviving good bacteria to multiply faster. It is widely used in pediatric medicine, where it has a strong safety profile and significantly reduces the duration of liquid stools in young children on antibiotics.
The Two-Hour Spacing Rule
If you choose a bacterial probiotic like LGG, timing matters. Taking it at the same time as your antibiotic is a mistake, because the drug will neutralize the probiotic in your stomach.
Follow the Two-Hour Rule: take your probiotic at least two hours before or two hours after your antibiotic dose. This spacing gives the probiotic enough time to pass through your stomach and reach your intestines safely, ensuring you actually get the benefit.
Probiotic Strains Compared: Efficacy for Preventing Diarrhea
| Organism | Strain | Type | Efficacy | Best Use |
| Saccharomyces | S. boulardii lyo CNCM I-745 | Yeast | Very High | Ideal during antibiotic course. Naturally immune to antibacterial drugs. |
| Lactobacillus | L. rhamnosus GG | Bacteria | High | Excellent for children. Restores gut barrier function during recovery. |
| Lactobacillus | L. acidophilus | Bacteria | Moderate | Good for general health. Susceptible to being killed by your prescription. |
| Bifidobacterium | B. infantis, B. lactis | Bacteria | Moderate | Best in multi-strain formulas post-treatment. Helps rebuild lower colon diversity. |
What to Eat When Antibiotics Cause Diarrhea

The Modified BRAT Diet
What you eat plays a huge role in how quickly you recover from dysbiosis. During active osmotic diarrhea, focus on foods that physically bind excess water. The classic recommendation is a modified BRAT diet: Bananas, Rice, Applesauce, and Toast.
These foods are bland, gentle on an inflamed stomach, and packed with a carbohydrate structure that slows digestion. You do not need to limit yourself exclusively to these four items. Boiled potatoes, plain oatmeal, and saltine crackers also work well. The goal is to eat easily digestible starches that give your struggling colon cells a break.
Soluble Fiber vs. Insoluble Fiber
Understanding the difference between the two types of dietary fiber is critical during recovery. Soluble fiber acts like a sponge inside your digestive tract, soaking up excess liquid and helping to firm up loose stools.
Insoluble fiber does the opposite. It sweeps through your intestines rapidly and speeds up bowel movements, which is the last thing you need during a bout of medication-induced diarrhea. Avoid raw vegetables, fruit skins, seeds, and tough leafy greens while symptomatic. Stick to soluble fiber sources like oats and peeled apples until your bowel movements return to normal.
Foods to Avoid During Dysbiosis
Certain foods will make things significantly worse. Dairy tops the list. When your gut lining is damaged, it temporarily loses the ability to produce lactase, the enzyme needed to digest milk. Consuming cheese, milk, or heavy cream will trigger severe gas, bloating, and explosive bowel movements.
Also avoid high-fat foods and greasy, fried meals. Fats require complex bile processing that your disrupted microbiome simply cannot handle. Finally, cut out refined sugars and artificial sweeteners. Sugar pulls more water into your bowel through osmosis, and artificial sweeteners like sucralose can be toxic to your surviving commensal bacteria.
Preventing Dehydration and Electrolyte Imbalance
The most immediate danger of persistent loose stools is not the dysbiosis itself. It is severe dehydration. When your colon stops absorbing water, you lose large amounts of fluid, sodium, and potassium.
Many patients make the mistake of reaching for sugary sports drinks. As noted above, sugar worsens osmotic diarrhea, making these drinks a dietary trap. Instead, sip clear broths to replace sodium, eat bananas for potassium, and drink plain water steadily throughout the day rather than gulping large amounts at once.
Making an Oral Rehydration Solution at Home
If symptoms become severe, you may need medical-grade hydration. Oral Rehydration Solutions (ORS) contain a scientifically balanced ratio of salt, water, and minimal glucose designed to force your intestines to absorb fluids. You can purchase pre-mixed ORS packets at any pharmacy. Look for brands that follow World Health Organization guidelines.
In a pinch, you can make an emergency ORS at home: mix one liter of clean water with exactly six level teaspoons of sugar and half a level teaspoon of salt. Stir until completely dissolved and sip slowly.
Who Is Most at Risk? Pediatric and Geriatric Concerns

Managing Symptoms in Children
Antibiotic-associated diarrhea affects different age groups in very different ways. In children, digestive upset is extremely common, especially since amoxicillin is so frequently prescribed for ear infections. The good news is that pediatric cases usually resolve quickly. Children have highly adaptable, resilient microbiomes that bounce back faster than adults.
When caring for young children, focus on preventing diaper rash. Osmotic diarrhea is acidic and can burn a toddler’s skin severely. Apply thick barrier creams containing zinc oxide after every bowel movement.
Why Older Adults Face Greater Danger
The picture is much more serious for older adults. Patients over 65 face a significantly higher mortality risk from secondary C. diff infections. As we age, the gut microbiome naturally loses diversity and resilience. An elderly microbiome has a much harder time recovering from chemical disruption, and the loss of short-chain fatty acids hits an older colon harder, leading to profound dehydration that can quickly cause kidney failure.
For patients over 65, using targeted probiotics is not optional. It is a critical standard of care that every prescribing physician should recommend.
Risks in Nursing Homes and Hospitals
Elderly patients in hospitals or nursing homes face the highest risk of all. These facilities are breeding grounds for C. diff spores, which can survive on bedrails, doorknobs, and medical equipment for months. C. diff is highly contagious and spreads through the fecal-oral route.
Importantly, alcohol-based hand sanitizers do not kill C. diff spores. Strict handwashing with warm soap and water is the only effective method. If you are visiting an elderly relative who is taking antibiotics, wash your hands thoroughly to protect their vulnerable digestive system.
Key Takeaways: Protecting Your Gut During Antibiotic Treatment
Antibiotics disrupt the delicate ecosystem of your gut microbiome. They wipe out the friendly commensal bacteria that keep digestion running smoothly. Without these beneficial microbes, your body stops producing short-chain fatty acids like butyrate, which halts colonic water absorption and leaves excess fluid trapped in your bowel. This mechanical failure results in osmotic diarrhea.
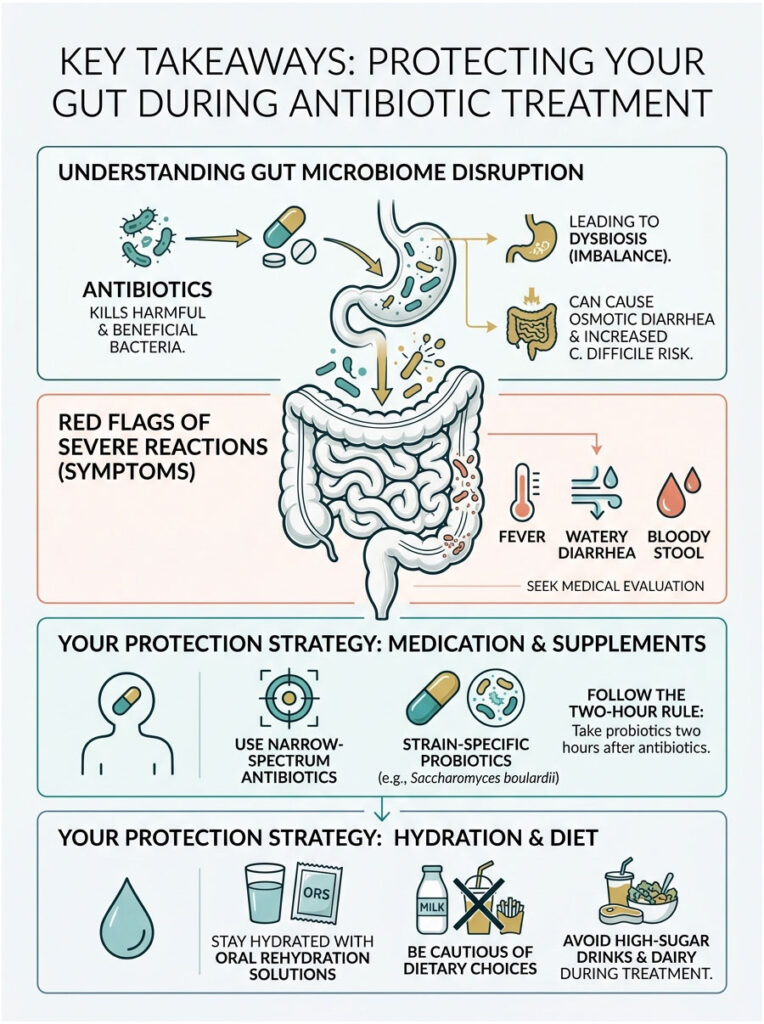
Beyond the discomfort, severe dysbiosis opens the door to dangerous opportunistic pathogens like Clostridioides difficile, which can cause life-threatening inflammation of the colon. Staying alert for red flags, such as severe watery diarrhea, fever, and bloody stool, can make the difference between a routine recovery and a medical emergency.
You have real tools at your disposal to protect yourself. Ask your doctor about narrow-spectrum antibiotics when possible. Use strain-specific probiotics like Saccharomyces boulardii and follow the two-hour spacing rule. Stay properly hydrated with oral rehydration solutions and support your surviving bacteria with soluble fiber.
Your digestive health is deeply connected to your overall immune system. The next time someone asks, “Do antibiotics cause diarrhea?” you will be able to explain exactly why it happens, and more importantly, how to prevent it.
Frequently Asked Questions
Why do antibiotics cause diarrhea even in healthy patients?
As a gastroenterologist, I explain this as ‘collateral damage.’ Antibiotics are often broad-spectrum, meaning they cannot distinguish between the pathogens causing your infection and the beneficial commensal bacteria in your gut. When these helpful microbes are killed, it leads to dysbiosis. This disruption stops the production of short-chain fatty acids like butyrate, which are essential for your colon to absorb water. When water absorption fails, it results in osmotic diarrhea.
How soon after starting a prescription will I notice digestive changes?
Most patients experience early-onset symptoms between 5 and 10 days after starting their antimicrobial therapy. This is the typical timeframe it takes for the medication to significantly deplete your internal ‘rainforest’ of bacteria below the threshold required for normal colonic function. However, in some high-risk cases, symptoms can begin within the first 48 hours.
Is it possible to develop diarrhea weeks after I finish my antibiotics?
Yes, this is a common clinical phenomenon known as delayed-onset diarrhea. You remain in a high-vulnerability window for up to 8 weeks after your last dose. This occurs because your microbiome and colonization resistance take significant time to rebuild. During this recovery phase, your gut remains fragile and susceptible to opportunistic pathogens like C. diff.
Which specific antibiotics carry the highest risk for gut issues?
In my clinical practice, I see the most severe disruptions with Lincosamides like Clindamycin and broad-spectrum Aminopenicillins like Augmentin. Cephalosporins and Fluoroquinolones (such as Ciprofloxacin) are also high-risk culprits. These drugs are particularly aggressive at clearing out the protective bacterial barriers in your large intestine.
How can I tell the difference between standard diarrhea and a C. diff infection?
Standard antibiotic-associated diarrhea is usually mild to moderate. However, Clostridioides difficile (C. diff) is a medical emergency. Red flags for C. diff include having more than five watery stools per day, a high fever, severe abdominal cramping, visible blood in the stool, and a characteristically foul, sickening odor. If you see these signs, seek immediate testing via a PCR or toxin assay.
Should I stop taking my medication if I develop loose stools?
You should never stop an antibiotic course prematurely without consulting your physician. Stopping early allows the remaining bacteria to mutate, leading to antibiotic resistance and the creation of ‘superbugs.’ Instead, contact your doctor to discuss switching to a narrow-spectrum alternative or adding medical-grade probiotics to manage the side effects.
What are the best probiotic strains for preventing antibiotic-associated diarrhea?
Not all probiotics are created equal. For antibiotic support, I recommend Saccharomyces boulardii, a medicinal yeast that isn’t killed by antibiotics, and Lactobacillus rhamnosus GG (LGG), which is excellent for patching the gut barrier. These specific strains have the strongest clinical evidence for reducing diarrhea risk by over 40%.
What is the ‘Two-Hour Rule’ for taking probiotics with antibiotics?
If you are using a bacterial probiotic (like LGG), you must space it at least two hours apart from your antibiotic dose. If you take them simultaneously, the antibiotic will simply kill the beneficial bacteria in the supplement before they reach your colon. Spacing them out ensures the probiotic survives long enough to provide therapeutic benefits.
What should I eat to help firm up my stool while on antibiotics?
I recommend a modified BRAT diet focusing on soluble fiber. Foods like bananas, white rice, applesauce, and peeled potatoes act like a sponge to soak up excess liquid in the bowel. Avoid insoluble fiber (like raw kale or seeds) and greasy foods, as these can irritate the already inflamed intestinal lining and speed up transit time.
Why should I avoid dairy products while dealing with dysbiosis?
When your gut lining is disrupted by antibiotics, it often suffers temporary damage to the brush border enzymes. This leads to a transient lactase deficiency, meaning your body cannot properly digest milk sugar. Consuming dairy during this time frequently triggers explosive gas, bloating, and worsening diarrhea.
Are sports drinks effective for rehydration during antibiotic-induced diarrhea?
Actually, I advise against high-sugar sports drinks. Excess sugar can pull even more water into the gut through osmosis, worsening the diarrhea. Instead, use a medical-grade Oral Rehydration Solution (ORS) which contains the precise ratio of salt and glucose needed to force the intestines to absorb fluids and prevent dangerous electrolyte imbalances.
Why is antibiotic-associated diarrhea more dangerous for patients over 65?
Older adults have a 10 times greater risk of severe complications because the geriatric microbiome is naturally less diverse and resilient. Seniors are much more likely to develop life-threatening dehydration, kidney failure, or recurrent C. diff infections. For this demographic, proactive microbiome protection with targeted probiotics is a mandatory standard of care.
Disclaimer
This article is for informational purposes only and does not constitute formal medical advice. The content is based on clinical observations and pharmacological data current at the time of publication. Always consult a qualified healthcare professional regarding your specific symptoms, medications, and treatment plans. Never discontinue prescribed antibiotics without direct medical supervision, as this can lead to antibiotic resistance.
References
- Centers for Disease Control and Prevention (CDC) – cdc.gov – Official data on Clostridioides difficile infection rates and prevention protocols in the U.S.
- American Gastroenterological Association (AGA) – gastro.org – Clinical guidelines on the use of probiotics for the prevention of antibiotic-associated diarrhea.
- Food and Drug Administration (FDA) – fda.gov – Safety communications and black box warnings regarding fluoroquinolone antibiotics and their side effects.
- World Health Organization (WHO) – who.int – Standardized guidelines for Oral Rehydration Solutions (ORS) and managing dehydration.
- Journal of Clinical Gastroenterology – “Saccharomyces boulardii: A Review of Efficacy” – Peer-reviewed research on the medicinal yeast’s impact on C. diff toxins.
- National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) – niddk.nih.gov – Detailed medical explanation of the gut microbiome and commensal bacteria functions.