After more than 15 years running CLIA-certified testing facilities, I get one question more than almost any other: “Which states in the USA don’t allow you to order your own blood tests?” It sounds simple enough. But the answer changes depending on where you live.
Table of Contents
Federal guidelines set baseline standards for testing accuracy across the country. However, individual state governments decide who can actually sign off on a laboratory requisition. That means your right to access your own biological data can vanish the moment you cross a state line.

This guide breaks down the specific medical gatekeeping laws in every category of state. I will walk through the clinical reasoning behind these rules, share real cost comparisons, and lay out the legally compliant workarounds available to patients who want to take charge of their health data right now.
Quick Answer
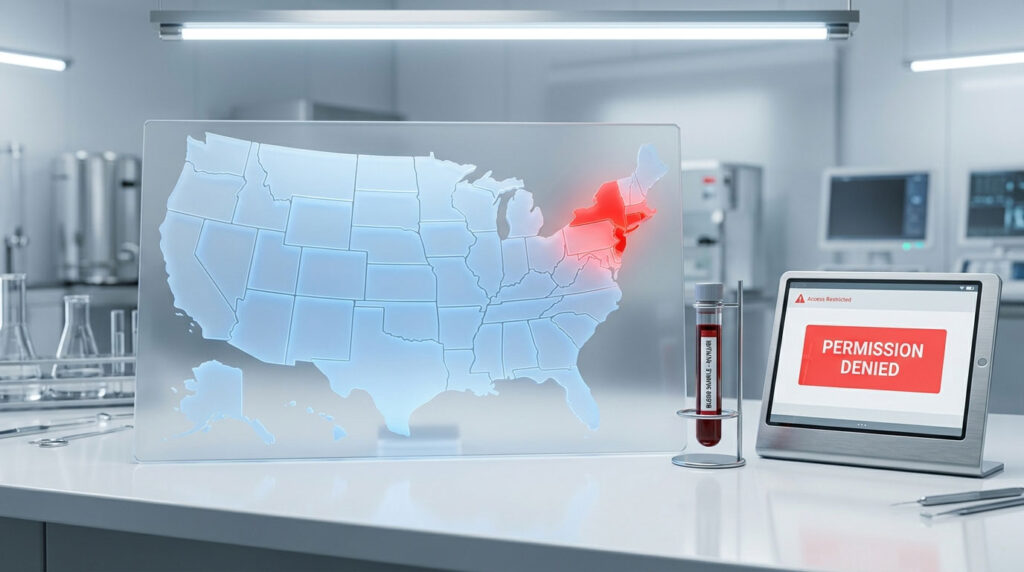
New York, New Jersey, and Rhode Island are the primary states that strictly prohibit direct consumer access to laboratory testing. In these states, every lab requisition needs a licensed healthcare provider’s signature. If you live in one of these three states, you cannot legally order your own diagnostic panels without going through a telehealth standing order or visiting an out-of-state facility.
Key Statistics on Consumer Testing in the U.S.
- 3 states maintain strict bans on consumer testing access (NY, NJ, RI)
- 37 states allow full or nearly unrestricted consumer testing access
- $2.4 billion is the projected market value of the self-pay lab industry by 2028
- 12 to 48 hours is the average turnaround time for results from accredited direct access labs
- Up to 70% potential savings on out-of-pocket costs compared to traditional hospital rates
- 92% accuracy correlation between traditional venous draws and modern home collection methods
What Is Direct Access Testing and How Does It Differ From Physician-Ordered Labs?
The medical testing industry is going through a major shift. We are moving away from reactive medicine and toward proactive screening. To make sense of this change, it helps to understand how the terminology works inside the laboratory.
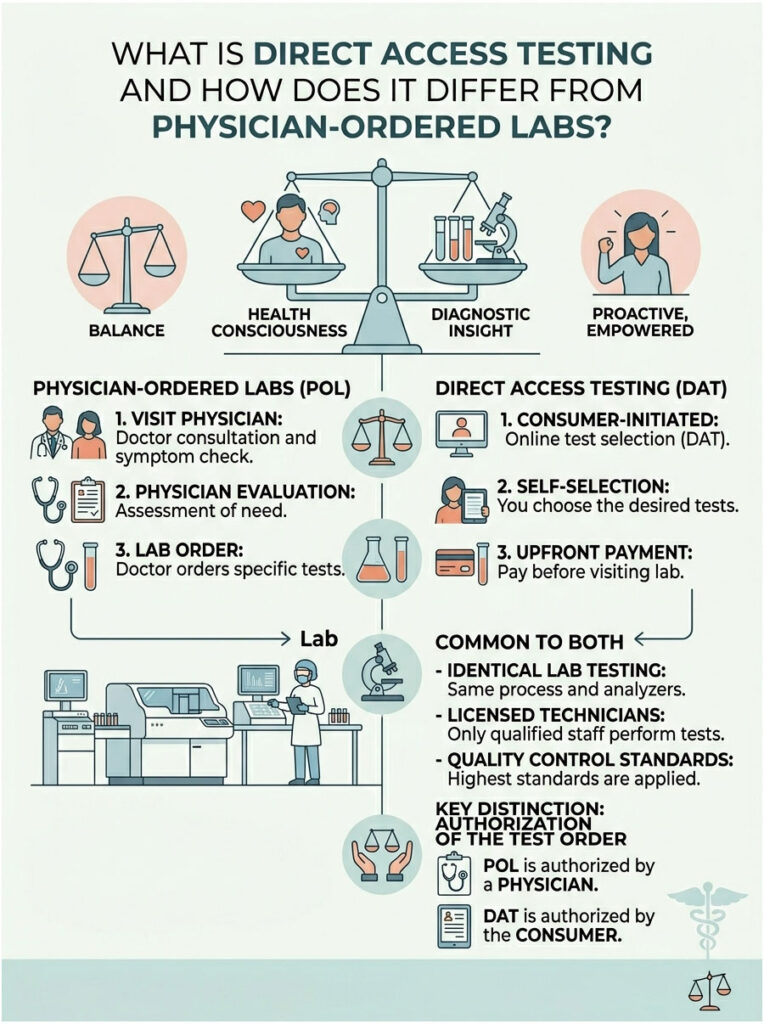
Direct Access Testing refers to lab tests that consumers can order on their own, without first consulting a physician. You may also hear it called consumer-initiated testing.
In the traditional model, you visit your doctor, they evaluate your symptoms, and then they write a lab order. You take that paper to a draw center and get your blood drawn. With direct access platforms, you skip the initial doctor visit altogether. You pick the tests you want online, pay upfront, and walk straight into a lab.
Here is the thing most people do not realize. The actual testing process inside the laboratory is identical no matter who ordered it. The same licensed technicians run the same samples on the same multi-million-dollar clinical analyzers. Your blood does not know whether a doctor or a website placed the order. The machines apply the exact same quality control standards either way. The only real difference is who authorized the test in the system.
Federal Oversight and CLIA Regulations
A common concern with self-ordered tests is whether they are safe and accurate. The short answer is yes, as long as the facility is properly regulated. The Centers for Medicare and Medicaid Services (CMS) oversees all laboratory quality in the United States through the Clinical Laboratory Improvement Amendments (CLIA).
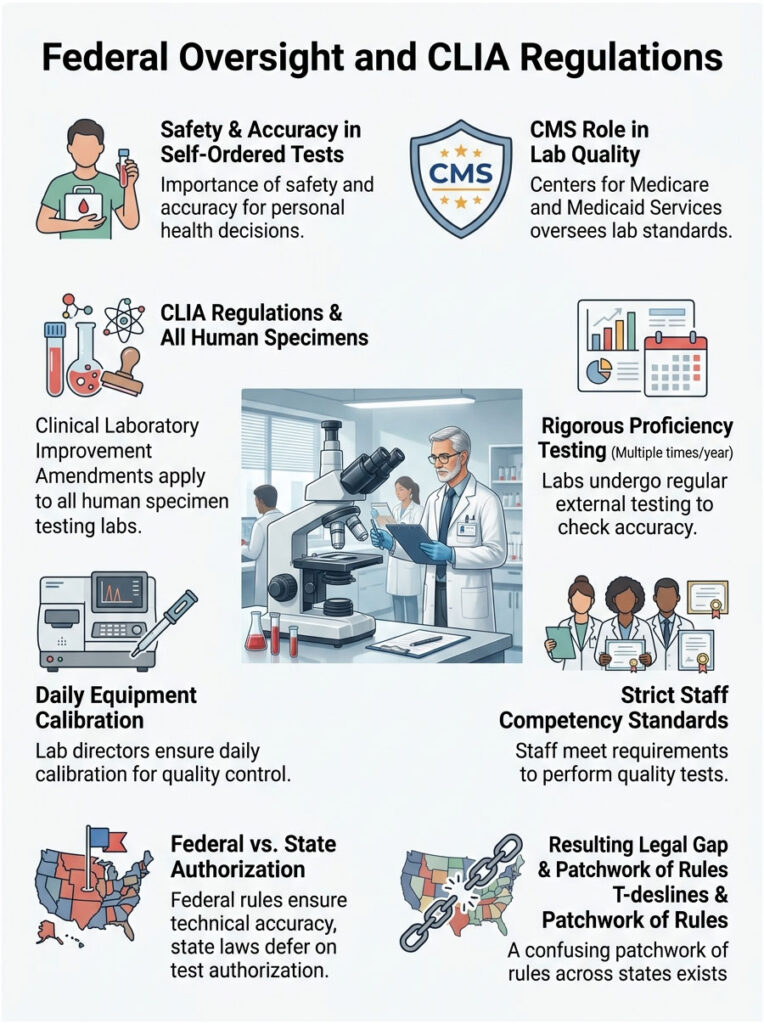
These federal regulations apply to every facility that tests human specimens for health assessment purposes. Regulated labs must pass rigorous proficiency testing multiple times a year. Directors must prove their equipment is calibrated daily and that their staff meets strict competency standards. Labs that fail these inspections get shut down.
But here is the catch. Federal rules guarantee technical accuracy, yet they defer entirely to individual state laws when it comes to who can authorize a test. The federal government explicitly states that national regulations do not override local state laws. This legal gap is what creates the confusing patchwork of rules we see across the country. A lab can be federally certified and still legally barred from serving patients in certain zip codes.
Why Consumer Tests Are Almost Always Self-Pay
You might wonder why you always have to pay cash for these tests. It comes down to how insurance billing works. Health insurance providers operate on a concept called medical necessity. To process a reimbursement claim, they require specific diagnostic codes known as ICD-10 codes. Only a licensed provider can generate one of those codes after evaluating your symptoms.
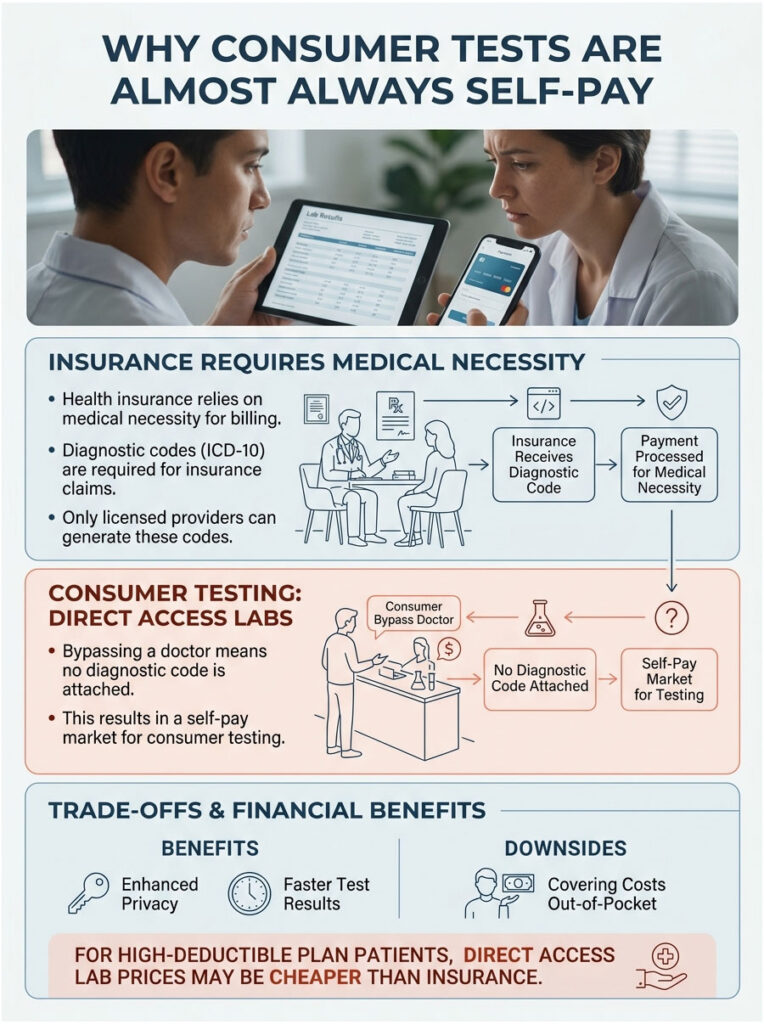
Because you are bypassing the doctor, no diagnostic code gets attached to the lab work. That makes consumer testing almost exclusively a self-pay market. You are choosing to step outside the traditional insurance system entirely.
The upside is incredible privacy and fast turnaround. The downside is that you cover the cost yourself. That said, for many patients on high-deductible plans, the cash price at a direct access lab is actually cheaper than what they would owe through insurance.
The Restricted States: Where You Cannot Order Your Own Blood Tests
Now let us get to the core question: which states in the USA don’t allow you to order your own blood tests? The Northeast contains the most restrictive regulatory environments in the country. If you live in one of these three states, your access to proactive health data is heavily controlled.
Legislators in these areas believe that medical data requires professional interpretation. They argue that handing patients raw biological data without clinical context is dangerous. As a result, they have built significant legal barriers against direct consumer access.
New York: The Most Restrictive State in America
Among laboratory professionals, New York is widely known as the hardest state to work in. The New York State Department of Health maintains extremely tight oversight over all clinical specimens through its Clinical Laboratory Evaluation Program (CLEP).
Under Section 570-581 of the Public Health Law, direct consumer access is explicitly banned. There are essentially no exceptions for routine wellness testing. You cannot simply walk into a lab and ask for a lipid panel.
Every single requisition must carry the physical or electronic signature of a licensed medical provider, whether that is an MD, DO, NP, or PA. Chiropractors and naturopaths are generally excluded from ordering complex panels in the state.
New York also aggressively restricts most out-of-state mail-in testing kits. Labs in other states face steep fines if they process specimens from a New York resident. The state actively audits shipping records to catch unauthorized kits crossing state lines.
The only narrow exceptions tend to involve specific public health initiatives, such as anonymous HIV testing designed to encourage vulnerable populations to seek help. Beyond that, the door to consumer autonomy is firmly shut.
New Jersey: Strict Physician Mandates
New Jersey follows closely behind New York in terms of restrictions. The state enforces strict gatekeeping policies for all diagnostic procedures. Local health department statutes require a direct clinician-patient relationship before any testing can take place.
Patient service centers operating in New Jersey face heavy and continuous scrutiny. State inspectors routinely audit these facilities to verify that every blood draw is tied to a valid, state-licensed physician’s order. If a lab technician draws blood without that order, they risk losing their phlebotomy certification. The facility itself can lose its operating license.
The state medical board says this system protects patients from unnecessary and expensive testing. Consumer advocates, on the other hand, argue it simply creates costly barriers to basic health information.
Rhode Island: Traditional Medical Gatekeeping
Rhode Island also heavily restricts consumer diagnostic autonomy. The state aligns with traditional medical models and legally requires a clinical evaluation before any phlebotomy services take place.
Ask any local laboratory director which states don’t allow you to order your own blood tests, and Rhode Island will always be on the list. The state treats lab testing as an exclusive tool for medical diagnosis, not a consumer wellness product.
Many Rhode Island residents express frustration with these rules. They just want to check their cholesterol or vitamin D without paying for a full doctor’s visit. Unfortunately, state law pushes them into the traditional healthcare system for even the most basic screening tests.
State-by-State Legal Status: Comparison Table
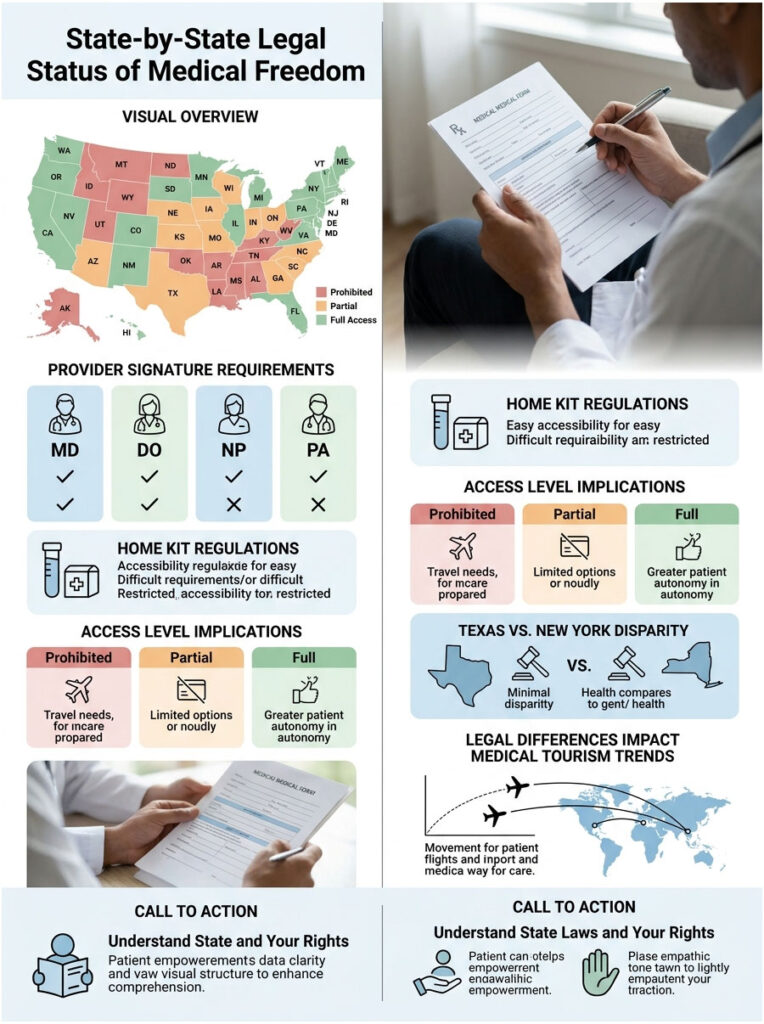
| State | Legal Status | Provider Signature? | Home Kit Rules | Access Level |
| New York | Prohibited | Yes (MD, DO, NP, PA) | Almost all banned | None |
| New Jersey | Prohibited | Yes | Highly regulated | None |
| Rhode Island | Prohibited | Yes | Heavily regulated | None |
| Maryland | Partial | Yes (complex panels) | Permitted (basic) | Limited |
| California | Partial | Yes (complex panels) | Mostly permitted | Limited |
| Arizona | Full Access | No | Fully permitted | Unrestricted |
| Texas | Full Access | No | Fully permitted | Unrestricted |
This table highlights the stark contrast between regions. A resident in Texas enjoys full medical freedom over their personal data. Meanwhile, a resident in New York must navigate layers of bureaucracy just to check their iron levels. This geographic gap fuels a growing market for medical tourism, with people routinely crossing state lines for basic preventive care.
Partial Access States: Limited Diagnostic Autonomy
Not every state operates in absolute terms. Several have adopted hybrid approaches, allowing residents to order specific low-risk tests while restricting access to complex diagnostic panels.
This middle ground tries to balance patient autonomy with medical safety, but it often creates confusion for both patients and lab staff. The rules are nuanced and change frequently based on state health board decisions.
California and Specific Test Allowances
California is a fascinating case study. The state permits consumers to order a specific list of basic wellness markers. You can walk into a lab and request a cholesterol check or a basic pregnancy test. The state Business and Professions Code outlines which screening tests are allowed, based on the logic that these markers do not require complex medical interpretation.
But the rules shift dramatically if you want a comprehensive hormone panel or heavy metals testing. California strictly restricts complex endocrine, genetic, or metabolic panels from direct consumer access. For those advanced tests, a doctor’s signature is mandatory. Many people assume they have full access in California, only to be turned away at the lab counter.
Maryland, Massachusetts, and Pennsylvania
These states also use hybrid models. Their health departments classify diagnostic tests into low-risk and high-risk categories. Low-risk tests generally include basic metabolic screening and STI testing, which the state views as public health priorities. High-risk tests cover genetic markers, complex cancer antigens, and esoteric autoimmune panels, all of which the state believes require professional interpretation.
If you are researching which states in the USA don’t allow you to order your own blood tests, these partial-access states sit right in the middle. They offer a taste of freedom but maintain strong regulatory guardrails.
The Clinical Reasoning Behind Medical Gatekeeping
Expert Insight: As a laboratory director, I see both sides of this debate every day. I strongly support patient autonomy, but laboratory data is highly nuanced. A single abnormal value does not always point to a disease. Context is everything in clinical pathology.
To understand why these restrictions exist, we need to look at the concerns medical boards raise. Lawmakers do not create these rules just to frustrate patients. They rely on guidance from professionals who have genuine worries about patient safety.
Diagnostic Validity and Clinical Context
Lab results are almost never black and white. A number on a page is not simply good or bad. Everything depends on the patient’s unique clinical context. Pre-analytical variables like fasting status, time of day, hydration, and supplement use all impact reference ranges significantly.
A trained physician understands how to weigh these factors. A patient might see an elevated glucose reading and panic, not realizing that their sweetened morning coffee caused a temporary spike. Ensuring diagnostic validity is the main reason medical boards insist on keeping physicians in the loop.
The Risk of Cyberchondria
There are real psychological risks when patients interpret complex data alone. On a routine Complete Blood Count or Comprehensive Metabolic Panel, it is very common to see one or two markers fall slightly outside the normal range.
In clinical practice, a mildly elevated liver enzyme might be completely harmless, possibly caused by an intense workout the day before. A doctor would recognize that and move on. But a patient reading their own results might search the internet, find worst-case scenarios, and convince themselves they have a serious illness. This phenomenon of internet-fueled health anxiety is known as cyberchondria. Gatekeeping policies aim to prevent that unnecessary distress.
Managing Critical Values and Emergencies
This may be the most serious concern for any lab director. When a test result shows an immediate, life-threatening condition, we call it a critical value. If a patient’s potassium is dangerously high, they could suffer a fatal cardiac event at any time.
In the traditional model, I call the ordering physician, who contacts the patient and directs them to the ER. With consumer-initiated testing, who do I call? The patient may not answer. Even if they do, they may not grasp the urgency. Managing critical values without a physician safety net is a real ethical and logistical challenge. Regulators point to this gap as a primary reason to restrict direct access.
Recent Legislative Shifts and the Rise of Patient Autonomy
Despite these concerns, the political tide is turning. Massive consumer demand for proactive healthcare is pushing state legislatures to reconsider their rules. The rise of high-deductible health plans has accelerated this trend. Patients are tired of paying steep hospital bills for basic preventive care, and they are lobbying their representatives to open up the lab market.
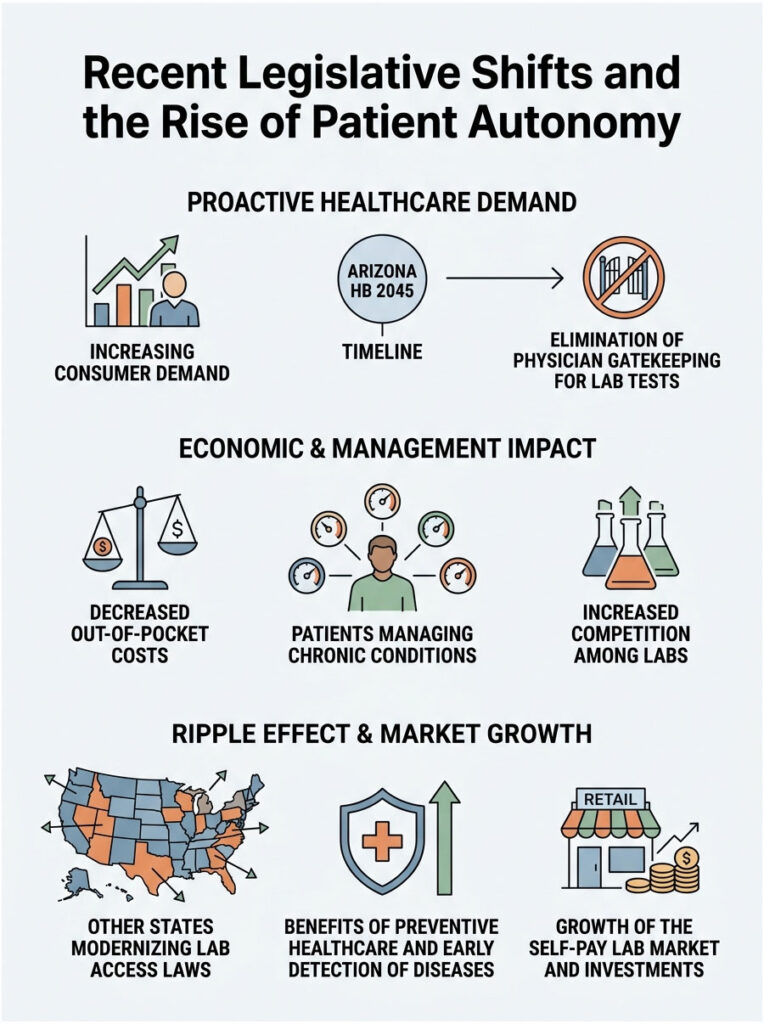
Arizona HB 2045: The Gold Standard for Patient Access
In 2015, Arizona completely disrupted the laboratory industry. The state passed House Bill 2045, which eliminated traditional physician gatekeeping for lab tests. Arizona lawmakers decided that citizens have a fundamental right to access their own biological data. Any consumer can now order any lab test from a certified facility without a doctor’s order.
The economic impact was immediate. Out-of-pocket testing costs dropped almost overnight as commercial labs began competing directly for consumer dollars. Transparency increased dramatically. Patients started taking active roles in managing chronic conditions like diabetes and thyroid disorders. The Arizona experiment proved that consumers can handle their own health data responsibly.
The Ripple Effect Across Progressive States
Arizona’s success created a powerful ripple effect. The list of states that don’t allow you to order your own blood tests is slowly shrinking. Florida, Colorado, Texas, and others have modernized their lab statutes in recent years.
These progressive states recognize that preventive healthcare saves money long-term. Letting citizens monitor their own lipid panels can reduce the burden of chronic disease. Early detection prevents expensive hospital stays. The self-pay lab market is booming in these permissive states, with major national labs investing heavily in retail storefronts.
Legal Workarounds for Residents of Restricted States
If you live in New York, New Jersey, or Rhode Island, you might feel locked out of modern healthcare. But the market has found legal ways to meet consumer demand. There are effective, fully compliant strategies you can use right now.
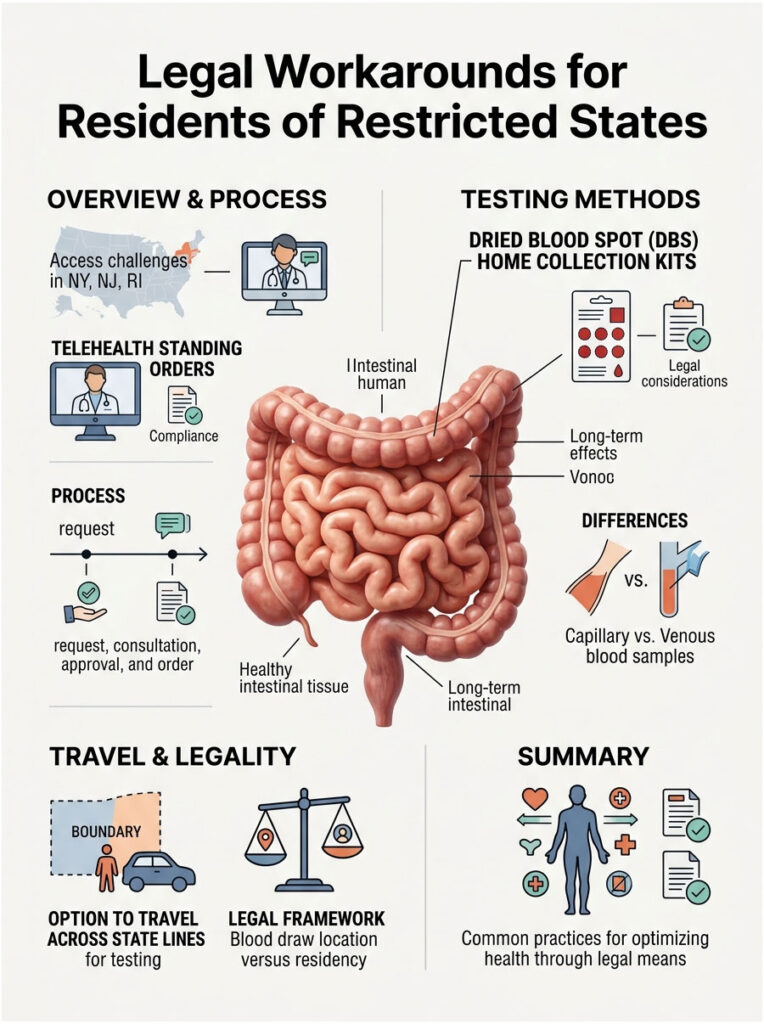
Telehealth Standing Orders
This is the most common and legally sound method in the industry today. Companies like Quest Diagnostics, LabCorp, and various digital health startups use what is known as an asynchronous telehealth standing order.
Here is how it works. You visit a direct-to-consumer lab website and pick the tests you want. You pay with a credit card. Behind the scenes, an independent state-licensed physician reviews your request, checking your age, gender, and test selections. If everything looks appropriate, the doctor signs the electronic requisition on your behalf.
Because a licensed physician officially authorized the test, it complies with local gatekeeping laws. You get the convenience of direct access, and the lab gets the legal compliance it needs. The cost of the physician review is usually bundled into your checkout price. You never actually speak with the doctor. The entire process runs through secure digital networks in the background.
Dried Blood Spot Home Collection Kits
Another popular workaround involves home collection technology. Many digital health companies offer Dried Blood Spot (DBS) kits that ship directly to your door. You prick your finger with a small lancet, place a few drops of blood on a specialized filter paper card, and mail it back in a prepaid envelope.
Regulatory loopholes sometimes apply to these kits. Because no formal phlebotomy service takes place in the state, the legal jurisdiction can get blurry. That said, states like New York still heavily restrict them and actively block shipments.
From a clinical standpoint, capillary blood from a finger prick is not exactly the same as venous blood from an arm draw. While the technology is improving quickly, traditional venous phlebotomy remains the gold standard for diagnostic accuracy.
Traveling Across State Lines
If you want a comprehensive venous draw but live in a restricted state, simple geography offers a solution. Many New York residents drive to Connecticut or Pennsylvania for testing. It is perfectly legal. The law applies to where the blood draw takes place, not where your driver’s license was issued. Once you cross the border, that state’s rules apply.
You can walk into a Pennsylvania lab, order your tests on a tablet, pay cash, and leave. Results arrive online within a few days. This is a common strategy for people deeply invested in optimizing their health.
Traditional Venous Draw vs. Home Collection vs. Telehealth: Comparison

| Feature | Venous Draw | Dried Blood Spot | Telehealth Order | Doctor Visit |
| Sample Quality | Gold standard | Variable | Gold standard | Gold standard |
| Legal in NY/NJ? | No (without MD) | Mostly no | Yes | Yes |
| Test Variety | Unlimited | Limited | Unlimited | Unlimited |
| Turnaround | 12-48 hours | 5-7 days | 24-48 hours | 24-48 hours |
| Physician Consult | Requires visit | Usually none | Included in fee | Required |
| Typical Cost | $$ | $ | $$ | $$$ |
The telehealth standing order comes out as the clear winner for most consumers. It combines the clinical accuracy of a venous draw with the legal compliance needed in restricted states. It is the ideal bridge between consumer demand and outdated legislation.
Real Cost Comparisons: Self-Pay vs. Insurance Billing
The traditional hospital billing system is notoriously opaque. A routine lipid panel billed through a hospital chargemaster can easily cost an insurance company $400 or more. If you are on a high-deductible plan, you might end up paying that entire inflated amount out of pocket.
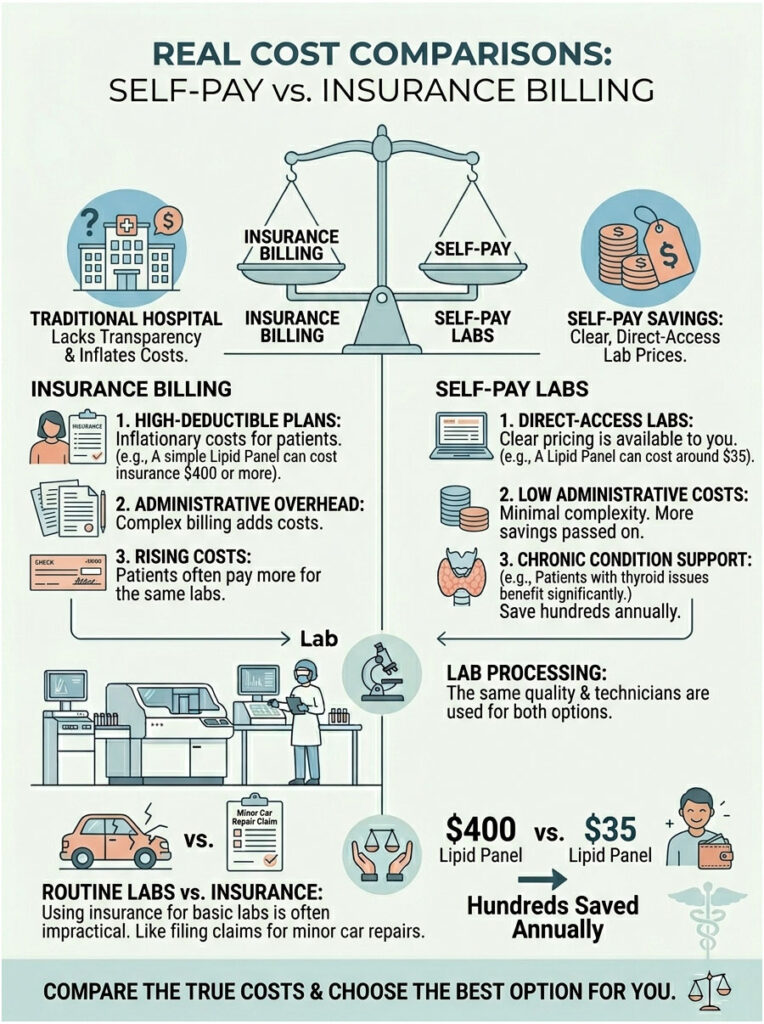
The self-pay lab market works very differently. That same lipid panel through a direct access portal typically costs around $35. The price drops dramatically because the massive administrative overhead of insurance billing is removed entirely.
Patients managing chronic thyroid conditions feel this difference acutely. They often need TSH levels checked quarterly. Using a direct access lab instead of insurance saves them hundreds of dollars a year. The financial reality is driving enormous consumer demand. For routine maintenance labs, using health insurance is like filing a car insurance claim for an oil change. It simply does not make financial sense.
Early Detection: How Consumer Testing Saves Lives
Beyond the financial savings, direct access testing genuinely saves lives through early detection. In our facilities, we see the clinical benefits of patient autonomy on a regular basis.
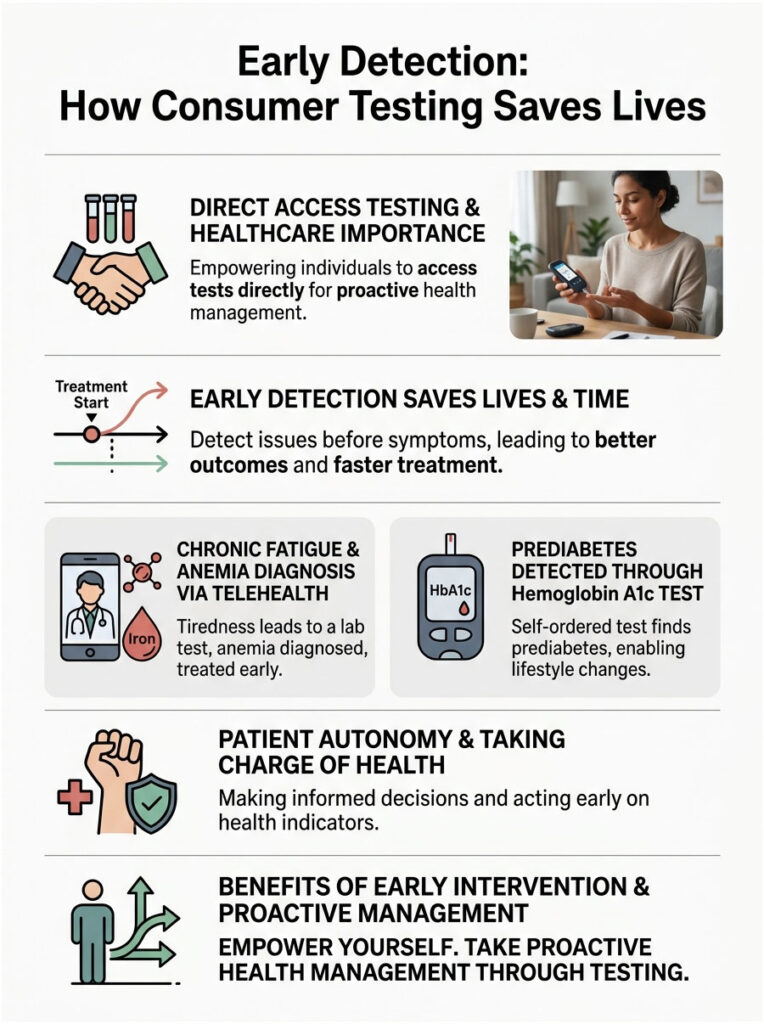
Consider someone who feels chronically fatigued but cannot get a doctor’s appointment for three months. They use a telehealth platform to order a basic anemia panel. The results reveal severe iron deficiency. By the time they finally see their doctor, they already have the data in hand, accelerating their treatment timeline significantly.
We also see frequent cases of early prediabetes detection. Patients who are simply curious about their health order a Hemoglobin A1c test. A slightly elevated result prompts them to change their diet well before they develop full-blown Type 2 diabetes. That kind of early intervention is exactly what preventive medicine is supposed to look like.
Privacy, Data Security, and HIPAA in Consumer Testing
When you handle your own healthcare, you also need to take responsibility for data privacy. Traditional medical records are protected by strict federal laws, but the picture is a bit more complex in the consumer testing space.
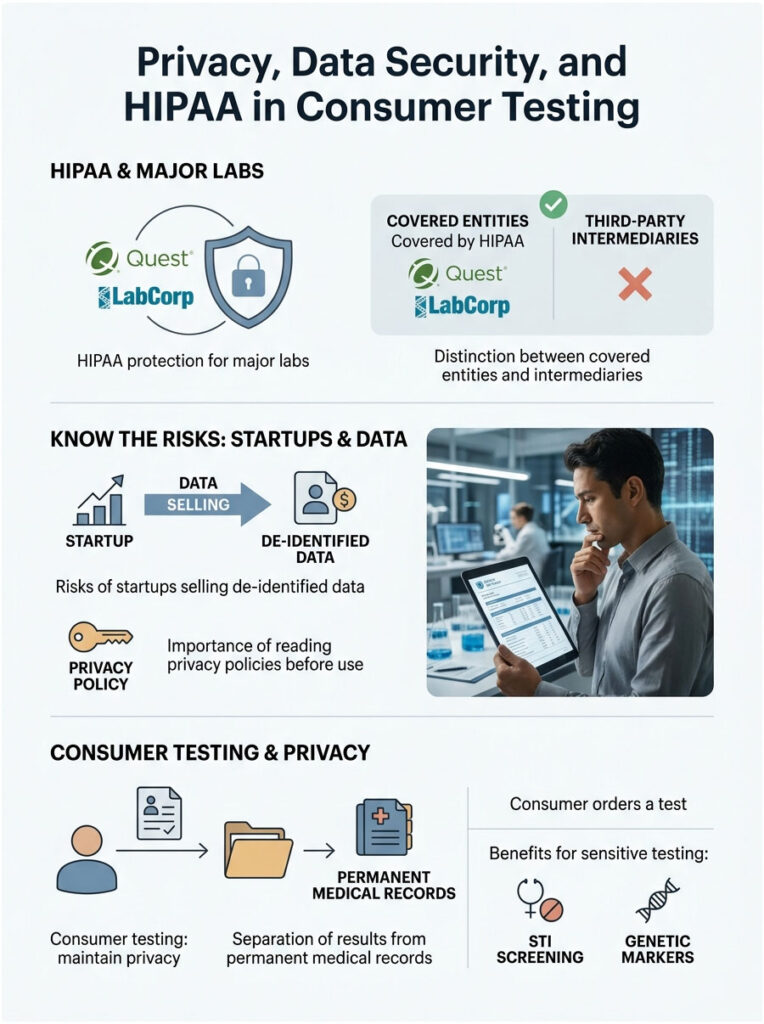
How HIPAA Applies to Direct Access Labs
If you use a major national laboratory like Quest or LabCorp, your data is fully protected by HIPAA. These organizations are classified as covered entities under federal law and must follow the same privacy standards as your local hospital.
However, some digital health startups operate as third-party intermediaries. They may not qualify as covered entities under the original wording of the law. Before using any consumer testing platform, read their privacy policy carefully. Look for specific language confirming that they will not sell your de-identified data to pharmaceutical companies. Some startups profit by aggregating and selling consumer health trends.
Keeping Results Off Your Permanent Medical Record
One of the biggest appeals of consumer testing is privacy. Many patients want to check their status for certain conditions without alerting a life insurance provider. Because you pay cash, the data stays completely separate from the traditional system.
No claims get filed to central clearinghouses. Results live securely in your private account with the testing company. The data only enters your permanent medical record if you choose to print the results and share them with your doctor. This privacy advantage is especially appealing for STI screening and for individuals checking genetic markers that could affect future insurance premiums.
The Future of Self-Pay Lab Testing and Proactive Healthcare
The laboratory industry is on the verge of a technological revolution. The days of waiting weeks for a doctor to interpret a basic blood test are coming to an end. Technology is democratizing biological data faster than ever.
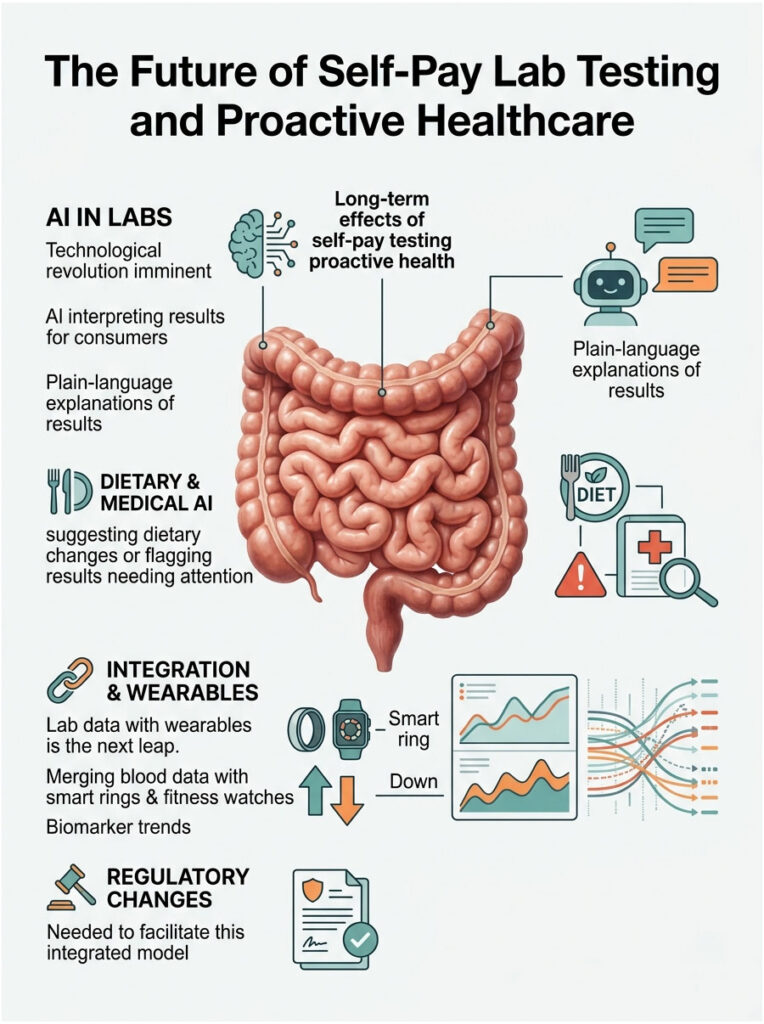
AI-Powered Result Interpretation
The biggest objection to consumer testing has always been data interpretation. Medical boards worry that patients will not understand their results. Artificial intelligence is addressing this problem head-on.
Modern testing platforms are integrating AI algorithms into their patient portals. When you receive your results, the AI analyzes your biomarkers against millions of data points and provides clear, plain-language explanations. It can suggest dietary changes or flag results that genuinely need a doctor’s attention. This technology bridges the gap between raw data and actionable medical guidance.
Integration With Wearable Technology
The next major leap involves merging blood data with wearable tech. Companies are already building platforms that combine lab results with data from smart rings and fitness watches.
Imagine an app that notices your resting heart rate climbing over a month. It automatically suggests a thyroid panel. You order the test with one click, get drawn the next day, and the app analyzes the results. This creates a closed-loop system of continuous health optimization. It removes the friction of the traditional healthcare model entirely. But this integrated future depends on states removing their restrictive gatekeeping laws.
Summary and Key Takeaways
Understanding which states in the USA don’t allow you to order your own blood tests is the first critical step in taking control of your health.
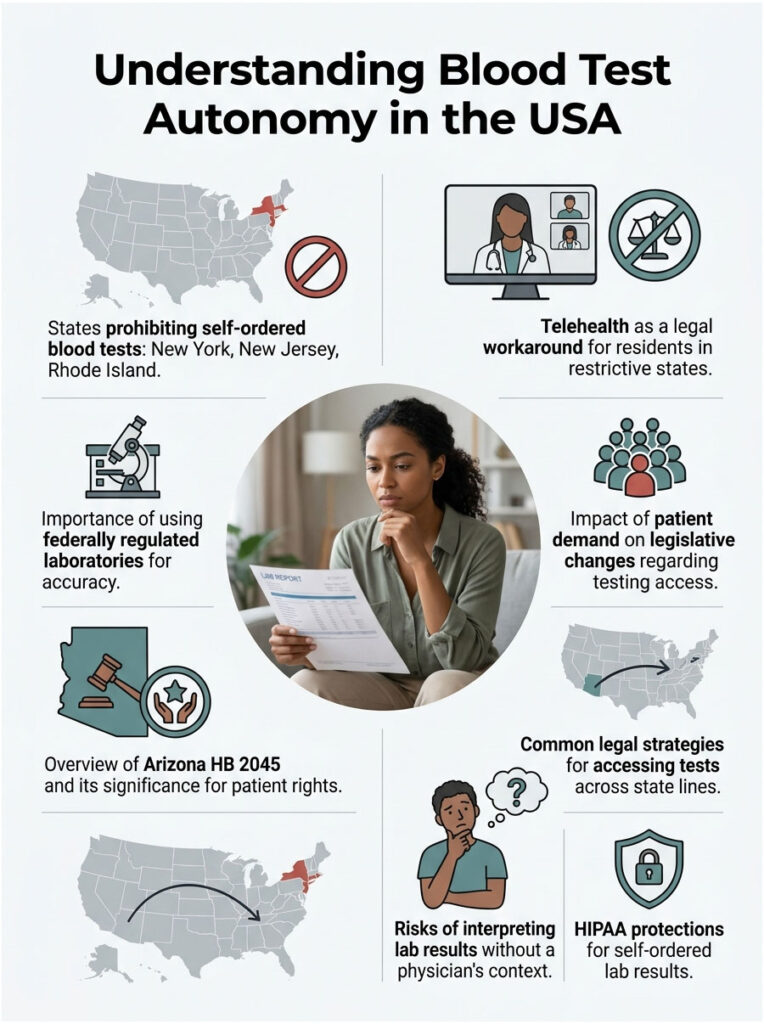
Geography dictates your diagnostic autonomy. The core restrictive states remain New York, New Jersey, and Rhode Island. In these locations, strict medical gatekeeping policies are firmly entrenched in the legal code.
For residents in restricted areas, telehealth networks with standing orders offer a safe, legal workaround. Always choose federally regulated laboratories to ensure the highest standard of accuracy.
The trend is clear: patient demand for transparent pricing and rapid access to health data will continue to force legislative change across the country. Whether you are in a fully open state or navigating restrictions, there are paths to the information you need. The key is knowing where you stand legally and using the right strategy for your situation.
Frequently Asked Questions
Which specific states currently prohibit Direct Access Testing (DAT) in 2026?
As a Laboratory Director, I closely monitor state statutes. Currently, New York, New Jersey, and Rhode Island maintain the strictest prohibitions against consumer-initiated testing. These states require a formal laboratory requisition signed by a state-licensed healthcare provider (MD, DO, NP, or PA) before any clinical specimen can be collected or processed.
Why does New York have such aggressive restrictions on home blood testing kits?
The New York State Department of Health operates the Clinical Laboratory Evaluation Program (CLEP), which is far more rigid than federal CLIA standards. They argue that without a clinician-patient relationship, there is no oversight for ‘pre-analytical variables’ or result interpretation. Consequently, they even ban most out-of-state mail-in kits to prevent residents from bypassing local gatekeeping laws.
Can I legally order my own lab tests if I live in a ‘Partial Access’ state like California?
In California, the law is nuanced. Under the state’s Business and Professions Code, you can order certain low-risk wellness markers, such as glucose or cholesterol, without a doctor. However, for more complex diagnostic panels involving hormones, genetics, or heavy metals, a physician’s signature is still legally mandated.
Are self-ordered blood tests processed differently than those ordered by a hospital?
Not at all. From my perspective inside the facility, the analytical process is identical. All specimens are run on the same high-throughput clinical analyzers by licensed technicians. Every lab must meet federal Clinical Laboratory Improvement Amendments (CLIA) standards, ensuring the same 92% or higher correlation regardless of the ordering party.
How do telehealth standing orders allow residents in restricted states to access testing?
This is a common legal workaround. When you purchase a test online, an asynchronous telehealth provider reviews your request. If appropriate, they issue a digital standing order. Because a licensed physician has technically authorized the requisition, it satisfies the ‘provider signature’ requirement in many jurisdictions, allowing the lab to legally process your sample.
Why won’t my health insurance cover tests that I order for myself?
Insurance reimbursement is predicated on ‘medical necessity,’ which requires specific ICD-10 diagnostic codes. Only a treating physician can legally assign these codes after a clinical evaluation. When you bypass the doctor, no ICD-10 code is generated, making the transaction a ‘self-pay’ event that falls outside the traditional insurance model.
What is the significance of Arizona HB 2045 for patient diagnostic autonomy?
Arizona’s House Bill 2045 is the gold standard for patient rights. It eliminated all physician gatekeeping, allowing citizens to order any laboratory assay they desire. This has fostered a highly competitive market, significantly lowering out-of-pocket costs and encouraging proactive screening for chronic conditions like diabetes and thyroid disease.
If I live in New Jersey, can I drive to a neighboring state to get blood work done?
Yes, this is a perfectly legal and common strategy. The law governs the location where the phlebotomy (blood draw) occurs, not your state of residence. Many residents of restricted states engage in ‘medical tourism’ by visiting patient service centers in permissive states like Pennsylvania or Connecticut to access direct testing.
How do laboratories handle ‘critical values’ for consumers who don’t have a primary doctor on file?
This is a major ethical concern in our industry. Most reputable direct-access platforms have a medical oversight team. If a result indicates a life-threatening emergency (a critical value), the platform’s physician network is responsible for contacting the patient immediately to direct them to emergency care.
What is the difference between a venous draw and a Dried Blood Spot (DBS) home kit?
A venous draw, performed by a phlebotomist, is the gold standard because it provides a high volume of stable serum or plasma. Dried Blood Spot kits use capillary blood from a finger prick. While DBS is convenient for home use, it can be subject to user error and is generally limited to a smaller menu of stable analytes compared to traditional venipuncture.
Are my self-ordered lab results protected by HIPAA privacy laws?
If you use major national laboratories (like Quest or LabCorp), your data is fully protected under HIPAA. However, some third-party tech startups may have different privacy tiers. Always review their policy to ensure they do not sell ‘de-identified’ biological data to pharmaceutical or marketing firms.
What are the clinical risks of interpreting my own lab data without a physician?
The primary risk is a lack of clinical context. A single ‘out-of-range’ marker may be benign depending on your hydration, fasting status, or recent exercise. Without a doctor to synthesize these variables, patients may experience ‘cyberchondria’—unnecessary anxiety driven by internet searches—or miss the subtle patterns that indicate true pathology.
Disclaimer
This article is for informational purposes only and does not constitute medical or legal advice. Laboratory regulations and state laws are subject to frequent change. Always consult with a qualified healthcare professional or legal expert before making decisions regarding diagnostic testing or interpreting medical results.
References
- Centers for Medicare & Medicaid Services (CMS) – Clinical Laboratory Improvement Amendments (CLIA) – Official federal oversight guidelines for all U.S. diagnostic facilities.
- New York State Department of Health – Clinical Laboratory Evaluation Program (CLEP) – Detailed statutes regarding laboratory specimen restrictions in New York.
- Arizona State Legislature – House Bill 2045 (2015) – The landmark legislation establishing full consumer access to laboratory testing in Arizona.
- American Association for Clinical Chemistry (AACC) – Direct-to-Consumer Testing Position Statement – Expert clinical perspective on the safety and validity of consumer-initiated labs.
- U.S. Department of Health & Human Services – HIPAA Privacy Rule – Official documentation regarding the protection of personal health information and laboratory data.
- Journal of Clinical Pathology – Research on Pre-analytical Variables – Peer-reviewed data on how fasting and hydration impact blood test accuracy.
- New Jersey Department of Health – Clinical Laboratory Improvement Services – Regulatory framework for diagnostic testing within the state of New Jersey.