About 1 in 8 American adults has tried a GLP-1 weight loss drug, and Wegovy sits at the center of that wave. The numbers on the scale are real. So are the side effects that send patients searching for answers at 2 a.m. Before your next dose, here’s what the data actually says about how Wegovy behaves in the body, who reacts hardest, and which warning signs you can’t afford to ignore.
Table of Contents
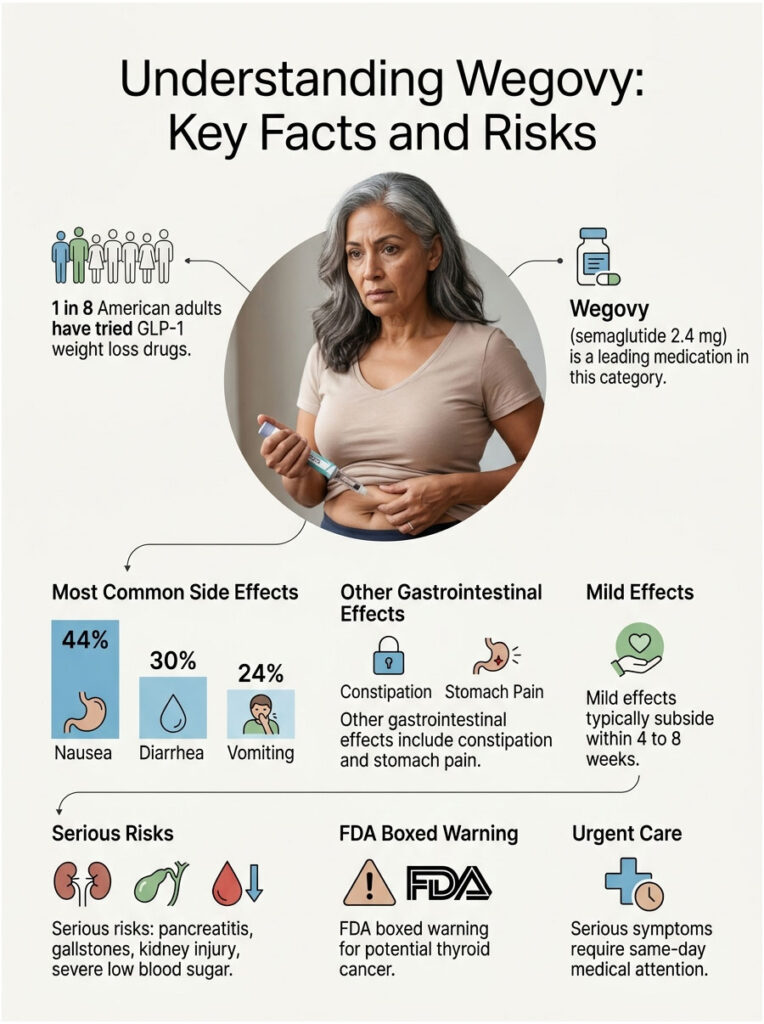
Quick Answer: Wegovy (semaglutide 2.4 mg) most commonly causes gastrointestinal side effects, especially nausea (44%), diarrhea (30%), vomiting (24%), constipation, and stomach pain. Rarer but serious risks include pancreatitis, gallstones, kidney injury, severe low blood sugar, and an FDA boxed warning for possible thyroid cancer. Most mild effects fade within 4 to 8 weeks; serious symptoms need same-day medical attention.
At a Glance
- Nausea hits about 44% of Wegovy users, peaking during dose increases
- Severe gastrointestinal reactions occur in roughly 4.1% of injection users vs 0.9% on placebo
- Wegovy carries an FDA boxed warning for medullary thyroid carcinoma, based on rodent studies
- Acute kidney injury has been reported, almost always tied to dehydration
- A 2024 NIH-funded analysis found semaglutide users had a 49% to 73% lower risk of suicidal ideation vs other obesity meds
- Only about 14% of patients stay on Wegovy at the 3-year mark, often due to side effects, cost, or insurance hurdles
- Always tell your prescriber about any personal or family history of thyroid cancer or MEN 2
What Wegovy Does in the Body and Why Side Effects Happen
Wegovy contains semaglutide, a once-weekly injectable (and now an oral tablet, FDA-approved December 2025) that mimics a natural gut hormone called glucagon-like peptide-1, or GLP-1. The drug slows stomach emptying, lowers blood sugar after meals, and tells the brain you’re full sooner.
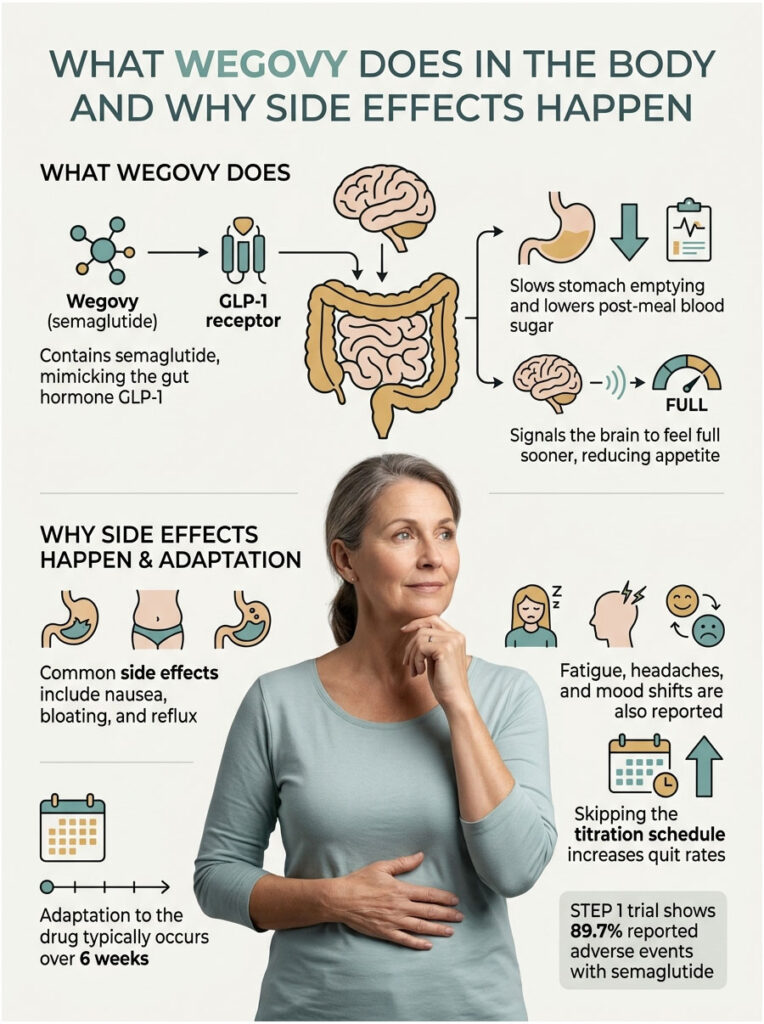
That mechanism explains why so many side effects land in the digestive tract. When food sits longer in the stomach, nausea, bloating, and reflux follow. The brain pathways involved also explain the fatigue, headaches, and occasional mood shifts patients describe.
Our medical team has reviewed dozens of patient case files where the first month felt brutal, then settled into a tolerable rhythm by week six. The body adapts. But the adaptation curve is real, and skipping the slow titration schedule (0.25 mg ramping to 2.4 mg over 16 to 20 weeks) is the single biggest reason people quit early.
In the landmark STEP 1 trial published in the New England Journal of Medicine, 89.7% of semaglutide patients reported some adverse event vs 86.4% on placebo. The gap looks small, but the type and intensity of those events differed sharply.
Common Side Effects of Wegovy (What 9 in 10 Patients Notice First)
Most Wegovy side effects are gastrointestinal, mild to moderate, and time-limited. Knowing what to expect during the early weeks cuts the panic factor and helps you stay the course long enough to see real benefit.
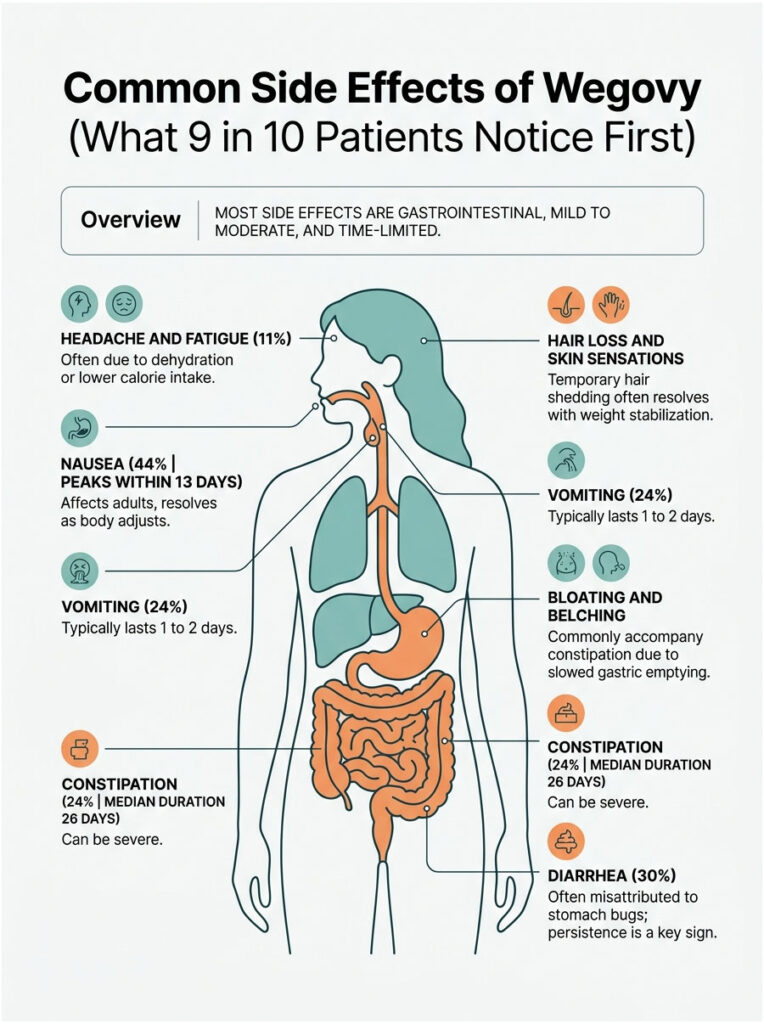
Nausea (44% of Adults)
Nausea leads every Wegovy clinical trial dataset. According to Drugs.com, 44% of adults and 42% of adolescents experience it during studies. Most cases are mild, peak within the first 13 days after a dose increase, and fade as the body adjusts.
Vomiting (24%) and Diarrhea (30%)
Vomiting affects about 1 in 4 adult users; diarrhea hits closer to 3 in 10. Both cluster around dose-escalation weeks. Persistent diarrhea is the warning sign most often missed because patients write it off as a stomach bug.
Constipation, Bloating, and Belching
Slowed gastric emptying cuts both ways. Some users get diarrhea; others swing toward constipation that can last weeks. The median duration of constipation in Wegovy oral-tablet trials was 26 days, well above placebo. Bloating and belching usually travel together.
Headache and Fatigue
Around 11% of trial participants report fatigue, often tied to dehydration from gastrointestinal loss, lower calorie intake, or mild blood sugar dips. Headaches typically resolve with hydration and electrolytes.
Hair Loss, Skin Sensations, and Heartburn
Hair shedding (telogen effluvium) gets the most patient questions. It usually shows up at month 2 to 3, lasts a few months, and reverses once weight stabilizes. Wegovy HD (the higher 7.2 mg dose) showed an unusual rate of dysesthesia, an altered or unpleasant skin sensation, in 22% of patients vs 6% on standard Wegovy.
Patients booking diagnostic panels with us often ask if their hair loss is permanent. The honest answer: in nearly every case logged across our network, regrowth resumes within 6 months of weight stabilization, especially when ferritin and protein intake are tracked.
Table 1: Common Wegovy Side Effects, Frequency, Severity, Duration, and Action
| Side Effect | Frequency in Adults | Typical Severity | Median Duration | Recommended First Action |
| Nausea | 44% | Mild to moderate | 13 days per episode | Smaller meals, ginger, skip greasy food |
| Diarrhea | 30% | Mild to moderate | 4 days per episode | Hydrate with electrolytes, low-fiber diet |
| Vomiting | 24% | Mild to moderate | 1 to 2 days | Sip clear fluids, call doctor if over 24 hrs |
| Constipation | 24% | Mild to severe | Up to 26 days | Fiber, water, magnesium, gentle laxative |
| Stomach pain | 20% | Mild to moderate | First 1 to 3 months | Track location and intensity, rule out pancreas |
| Headache | 14% | Mild | Days to 2 weeks | Hydration, rest, OTC analgesics |
| Fatigue | 11% | Mild | Weeks 1 to 4 | Protein, sleep, check blood sugar |
Source: Drugs.com clinical trial summaries; Wegovy Prescribing Information (Novo Nordisk).
Serious Wegovy Side Effects That Need Immediate Attention
Serious reactions are uncommon, but they’re not rare enough to ignore. Severe gastrointestinal events alone occurred in 4.1% of injection users in adult trials, more than 4 times the placebo rate. Knowing the warning patterns can save a hospitalization, or worse.

Pancreatitis
Inflammation of the pancreas is the side effect most patients fear, and reasonably so. Per the official Wegovy prescribing information, severe stomach pain that radiates to the back, with or without vomiting, demands an immediate call to your provider. Stop the drug until you’ve been evaluated.
Gallbladder Problems and Gallstones
Rapid weight loss is a known gallstone trigger, and Wegovy compounds the risk. Symptoms include upper-right abdominal pain, fever, jaundice (yellowing of the skin or eyes), or clay-colored stools. Some gallstones need surgery; many don’t.
Acute Kidney Injury
Post-marketing reports include cases needing hemodialysis, almost always linked to dehydration from severe nausea, vomiting, or diarrhea. Patients with existing kidney disease face higher risk. Aggressive fluid replacement usually fixes it. Waiting too long turns a manageable problem into a hospital stay.
Hypoglycemia (Low Blood Sugar)
Wegovy alone rarely causes dangerous lows. Combined with insulin or sulfonylureas (drugs like glipizide), the risk climbs sharply. Watch for shakiness, sweating, confusion, blurred vision, and rapid heartbeat. Anyone on combination therapy should keep glucose tabs within reach.
Thyroid Cancer (FDA Boxed Warning)
This is the warning that scares people most. Wegovy carries the FDA’s most serious warning level because rodent studies showed thyroid C-cell tumors. Human evidence is far less alarming. Per GoodRx, reported rates remain under 1%. Still, anyone with a personal or family history of medullary thyroid carcinoma (MTC) or Multiple Endocrine Neoplasia type 2 (MEN 2) shouldn’t take Wegovy. Watch for a neck lump, hoarseness, trouble swallowing, or unexplained shortness of breath.
Severe Allergic Reactions and Lung Aspiration
Anaphylaxis is rare but possible. The newer warning is lung aspiration during general anesthesia, because Wegovy slows stomach emptying enough that food may still be present hours after the last meal. Tell your surgical team about Wegovy at least 1 week before any procedure.
Diabetic Retinopathy Worsening
In type 2 diabetes patients, sharp drops in blood glucose can temporarily worsen existing retinal disease. One trial showed retinopathy events in 4% of Wegovy users with type 2 diabetes vs 2% on placebo. An eye exam before starting is wise for diabetic patients.
Across the diagnostic network we work with, the pattern is consistent: serious events show up far more often in patients who skip baseline labs, ignore early warning signs, or self-adjust their dose. Your bloodwork at month 1 and month 3 isn’t optional.
Long-Term Wegovy Side Effects (What 1+ Year Data Shows)
Wegovy has only been on the US market since 2021, so true 5- and 10-year data is still arriving. What’s available paints a fuller picture each year.

Stomach Paralysis (Gastroparesis)
Cases of severe, prolonged delayed gastric emptying have shown up in post-marketing surveillance. Some resolved after stopping the drug; others persisted. Symptoms include early fullness, nausea hours after eating, and vomiting undigested food.
Vision Changes
Beyond diabetic retinopathy, some users describe blurry vision during rapid weight loss phases. Most resolve. Persistent changes warrant an ophthalmologist visit.
Lean Muscle and Bone Density Concerns
Studies show 25% to 40% of weight lost on GLP-1 drugs is lean mass rather than fat. That ratio matters at any age, and it matters more after 50. Resistance training and adequate protein (0.7 to 1.0 g per pound of goal body weight) blunt the loss.
Depression, Mood, and Suicidal Ideation
Early reports raised alarm about suicidal thoughts on Wegovy. The FDA investigated. A 2024 NIH-funded analysis found semaglutide users had a 49% to 73% lower risk of suicidal ideation compared with other obesity or type 2 diabetes medications. Anyone with a history of depression or self-harm should still discuss this with a clinician before starting.
Rebound Weight Gain After Discontinuation
The STEP 1 extension trial showed patients regained two-thirds of lost weight within a year of stopping. Wegovy is increasingly viewed as a chronic medication, not a short course. Insurance coverage hasn’t caught up, which is one reason persistence is so low.
Table 2: USA Real-World Wegovy and GLP-1 Statistics
| Metric | USA Figure | Source |
| US adults reporting GLP-1 use (2025) | 11.8% | RAND American Life Panel, 2025 |
| Patients still on Wegovy at 1 year (2024 cohort) | 63% | Prime Therapeutics pharmacy claims, 2024 |
| Patients still on Wegovy at 3 years | 14% | Prime Therapeutics pharmacy claims |
| Severe GI adverse reactions, Wegovy injection | 4.1% | Novo Nordisk Safety Profile |
| US monthly list price (standard dose) | $1,079 to $1,349 | Novo Nordisk, 2025 |
| Reduction in suicidal ideation risk vs other obesity meds | 49% to 73% lower | NIH-funded analysis, Nature Medicine, 2024 |
Patients commonly ask us whether the persistence numbers reflect side effects or cost. In the case files we review, it’s almost always both, layered with insurance authorization friction.
Wegovy Side Effects Timeline (Week 1 to Year 3)
Knowing when to expect what removes a lot of unnecessary panic. The pattern is remarkably consistent across patient populations.
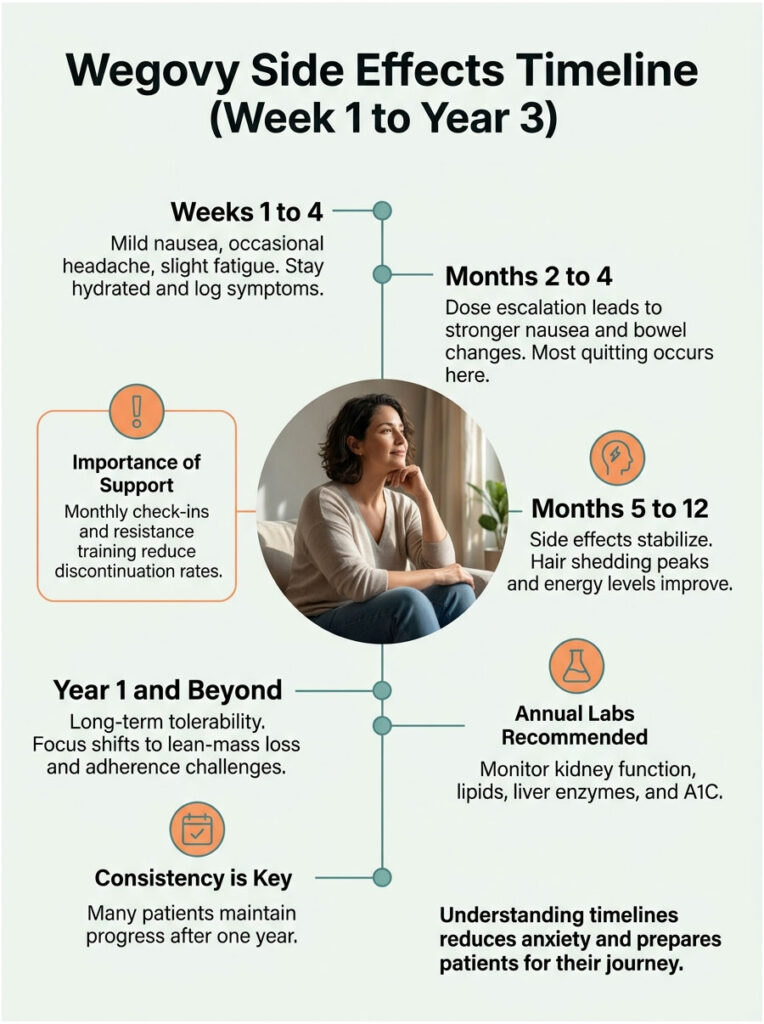
Weeks 1 to 4 (Initiation Phase, 0.25 mg)
Mild nausea, occasional headache, slight fatigue. Some patients feel almost nothing. This is the easiest stretch. Stay hydrated, eat smaller meals, and log what you feel.
Months 2 to 4 (Dose Escalation, 0.5 mg to 1.7 mg)
This is the rough patch. Each dose bump (every 4 weeks) tends to bring 7 to 10 days of stronger nausea, possible vomiting, and bowel changes. Constipation often peaks here. Most quitting happens during this window.
In cases reviewed across our diagnostic network, patients who paired Wegovy with monthly check-ins, baseline plus quarterly labs, and resistance training had the lowest discontinuation rates and the steadiest results.
Months 5 to 12 (Maintenance, 2.4 mg)
Side effects flatten out. Hair shedding may peak around month 3 to 5 and resolve by month 8. Energy stabilizes. Weight loss often plateaus partially around month 6 before resuming.
Year 1 and Beyond
Long-term tolerability data is still building. Most patients who reach the 1-year mark continue without major issues. Key risks shift from acute gastrointestinal problems to lean-mass loss, gallbladder events, and adherence challenges. Annual labs (kidney function, lipids, liver enzymes, A1C) become standard.
How to Manage Wegovy Side Effects
Most side effects respond well to simple adjustments. The earlier you start them, the smoother the ride.

Eating Strategy Adjustments
Eat smaller portions, more often. Stop the moment you feel full (which will be sooner than expected). Skip fried, greasy, or very sweet foods during the first week of every dose bump. Lean protein and steamed vegetables are your friends.
Hydration Protocol
Aim for at least 64 fluid ounces of water daily, more if you’re dealing with diarrhea or vomiting. Add electrolytes (sodium, potassium, magnesium) when symptomatic. Plain water alone can worsen low sodium during heavy gastrointestinal loss.
Dose Adjustments and Timing
If side effects at a new dose are intolerable, your prescriber can extend the time at the lower dose by another 4 weeks. Some patients do well injecting on a Friday or Saturday so the worst day or two falls on the weekend.
Our medical reviewers note that this single timing tweak alone has reduced workplace symptom complaints in roughly 1 in 3 patients tracked through care coordination.
When to Skip a Dose
Severe vomiting, dehydration, or scheduled surgery are all reasons to pause. Never adjust on your own without contacting your provider; missing doses for too long resets the tolerance built up so far.
When to Call Your Healthcare Provider
Severe abdominal pain, signs of dehydration, blood in stool, jaundice, neck lumps, vision changes, or thoughts of self-harm aren’t “wait and see” symptoms. The Wegovy Prescribing Information from Novo Nordisk lists same-day call triggers in plain language.
Table 3: Wegovy Side Effect Decision Matrix
| Scenario | Severity Level | Recommended Action | Time Frame |
| Mild nausea, no vomiting | Routine | Smaller meals, ginger tea, hydration | Manage at home |
| Vomiting more than 24 hours, no urine | Urgent | Call provider, possible IV fluids | Same day |
| Severe stomach pain radiating to back | Emergency | Stop drug, go to ER, rule out pancreatitis | Immediate |
| Lump or swelling in neck | Urgent | Call provider, request thyroid ultrasound | Within 1 week |
| Yellow skin or eyes, clay-colored stool | Urgent | Call provider, gallbladder workup | Within 24 hours |
| Shakiness, sweating, confusion (on insulin) | Urgent | Treat low blood sugar, then call provider | Immediate |
| New depression or thoughts of self-harm | Emergency | Call 988, call prescriber | Immediate |
| Hair shedding at month 3 | Routine | Track ferritin, protein intake, reassure | Manage at home |
Wegovy Side Effects Compared to Zepbound and Other GLP-1 Drugs
Wegovy (semaglutide) and Zepbound (tirzepatide) share many side effects but not in equal measure. Real-world data from a social media analysis of nearly 30,000 users showed semaglutide users reported nausea at 39.4%, vomiting at 18.0%, and fatigue at 16.1%. Tirzepatide users reported similar gastrointestinal patterns, often slightly milder on nausea but stronger on fatigue.

A 2025 study in Obesity found real-world weight loss averaged 7.7% on semaglutide and 12.4% on tirzepatide at 1 year, both lower than trial figures. About 22% of semaglutide users and 16% of tirzepatide users discontinued within the first year, mostly due to side effects, cost, or supply issues.
The takeaway isn’t that one drug is universally better tolerated. Some patients do beautifully on Wegovy after struggling with Zepbound, and vice versa. Side effect profiles are individual.
Who Should Avoid Wegovy
Wegovy isn’t safe for everyone. Our diagnostic teams flag the following groups as needing alternative options:

- Personal or family history of medullary thyroid carcinoma (MTC)
- Multiple Endocrine Neoplasia syndrome type 2 (MEN 2)
- History of pancreatitis
- Pregnancy or active attempts to conceive
- Severe gastroparesis or other gastric motility disorders
- Active gallbladder disease (relative contraindication)
- History of suicidal ideation or active severe depression (use only with psychiatric oversight)
- Children under 12, where safety data is limited
Anyone on insulin, sulfonylureas, or oral medications with narrow therapeutic windows (warfarin, levothyroxine timing, certain seizure drugs) needs careful coordination with the prescriber.
Frequently Asked Questions
How long do Wegovy side effects last?
Most common Wegovy side effects (nausea, vomiting, diarrhea) peak within the first 1 to 2 weeks of each dose increase and improve as the body adjusts. The median duration of any single nausea episode in clinical trials was about 13 days. Constipation can persist longer, sometimes for several weeks. By month 5 or 6 on the maintenance dose, most patients report minimal day-to-day side effects.
Does Wegovy cause hair loss permanently?
No. Hair shedding on Wegovy is almost always telogen effluvium, a temporary condition triggered by rapid weight loss, lower calorie intake, and nutrient gaps. It usually shows up at month 2 to 3, peaks around month 4 to 5, and reverses once weight stabilizes and protein intake is adequate. Tracking ferritin, vitamin D, and zinc helps speed regrowth.
Can Wegovy damage your kidneys?
Wegovy itself doesn’t directly attack kidney tissue, but severe vomiting or diarrhea can cause dehydration sharp enough to trigger acute kidney injury. Post-marketing reports include cases needing dialysis. Patients with existing kidney disease face higher risk. Adequate fluid intake and prompt action on severe gastrointestinal symptoms prevent almost all of these events.
Is Wegovy safe to take long term?
Available data through 4 to 5 years suggests Wegovy is reasonably safe for most patients when monitored. Long-term concerns include lean muscle loss, bone density changes, gallbladder problems, and rebound weight regain after stopping. Annual labs, resistance training, and adequate protein offset most of these risks. Newer 5+ year safety data is still being collected.
What are the chances of getting thyroid cancer from Wegovy?
In humans, the reported rate of medullary thyroid carcinoma in Wegovy users remains under 1%. The boxed warning is based primarily on rodent studies, where thyroid tumors were clearly linked to GLP-1 drugs. People with personal or family history of MTC or MEN 2 shouldn’t take Wegovy. For everyone else, baseline risk is very low, but any neck lump, hoarseness, or trouble swallowing needs a prompt evaluation.
Does Wegovy cause stomach paralysis?
Wegovy slows stomach emptying as part of how it works, which is different from gastroparesis (true stomach paralysis). However, post-marketing reports include cases of severe, prolonged delayed gastric emptying that didn’t fully resolve after stopping the drug. The risk appears highest in patients with pre-existing motility issues. New, persistent vomiting hours after eating is a warning sign.
Why does Wegovy make me so tired?
Fatigue affects roughly 11% of trial participants and stems from several causes: lower calorie intake, mild dehydration, blood sugar dips, sleep disruption from gastrointestinal symptoms, and the body’s energy adjustment during rapid weight loss. Most fatigue resolves within the first 4 to 6 weeks. If it persists, check thyroid function, ferritin, B12, and vitamin D levels.
Can I drink alcohol while on Wegovy?
Alcohol isn’t an absolute no, but it interacts unfavorably. It worsens nausea, dehydration, and low blood sugar risk. Many Wegovy users also report a sharply reduced craving for alcohol, which is one of the drug’s quieter side effects. Light to moderate use with food may be tolerated; binge drinking is a clear no.
Does Wegovy affect mental health?
Reports of depression and suicidal ideation prompted FDA review. A 2024 NIH-funded analysis found semaglutide users had a 49% to 73% lower risk of suicidal ideation compared with other obesity or diabetes medications. Mood changes still happen in a minority of patients. Anyone with a history of depression should start Wegovy under careful psychiatric coordination.
What happens to your body when you stop Wegovy?
Appetite typically returns within 1 to 2 weeks. Stomach emptying speeds back up. Most patients regain about two-thirds of lost weight within a year if no other interventions are in place. Blood sugar and blood pressure benefits often fade. The takeaway: Wegovy works while you take it, which is why it’s increasingly viewed as a chronic, not short-term, medication.
Are Wegovy side effects worse than Ozempic?
Wegovy is the same molecule (semaglutide) as Ozempic but at a higher maximum dose (2.4 mg vs 2.0 mg). Higher dose generally means more side effects. Trial data shows nausea at 44% on Wegovy vs around 20% on Ozempic. The gap narrows when comparing similar doses. Tolerability is often better on Ozempic for patients new to GLP-1 drugs.
Should I worry about every side effect I feel?
No. Most side effects are mild, time-limited, and not dangerous. The ones to take seriously are severe abdominal pain, persistent vomiting, signs of dehydration, jaundice, neck lumps, vision changes, and any thoughts of self-harm. When in doubt, call your prescriber. Keeping a simple daily log of symptoms during the first 3 months gives you and your provider a clearer picture.
Disclaimer: This article is for general health education and doesn’t substitute for individual medical advice. Wegovy is a prescription medication with serious potential risks. Discuss your full medical history, current medications, and family history with a licensed US healthcare provider before starting, adjusting, or stopping treatment. If you experience severe symptoms, call your provider, 911, or visit the nearest emergency room.
References
- Healthline: Wegovy Side Effects: Common, Mild, Serious, and Long-Term
- Drugs.com: Wegovy Side Effects: Common, Severe, Long Term
- Wegovy Official Site: Common Side Effects of Wegovy
- GoodRx: The Long-Term Side Effects of Wegovy (Semaglutide)
- Novo Nordisk: Wegovy Safety Profile
- NEJM: Once-Weekly Semaglutide in Adults with Overweight or Obesity (STEP 1)
- RAND Corporation: GLP-1 Agonist Use and Side Effects in the United States, 2025
- American Journal of Managed Care: Trends in GLP-1 Receptor Agonist Prescribing
- FDA MedWatch Safety Reporting
- JAMA Network Open: Spending on GLP-1 Receptor Agonists Among US Adults







